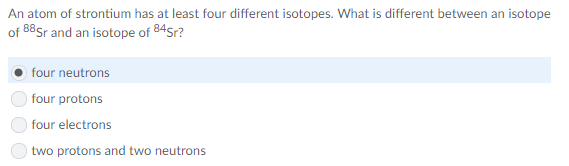Chemistry
15 Online
 rootbeer003:
rootbeer003:
help with these questions
Join the QuestionCove community and study together with friends!
Sign Up
 rootbeer003:
rootbeer003:
 rootbeer003:
rootbeer003:
@Elsa213 @HuskyNation @Ultrilliam @Vocaloid
 rootbeer003:
rootbeer003:
@dude @ThisGirlPretty
 Vocaloid:
Vocaloid:
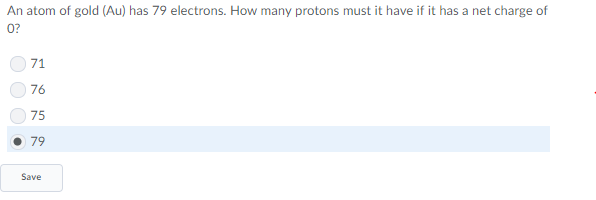
in order to be neutral, number of electrons = number of protons
re-read the problem and see what you can get from this
 rootbeer003:
rootbeer003:
i still dont know
Join the QuestionCove community and study together with friends!
Sign Up
 Vocaloid:
Vocaloid:
gold has 79 electrons according to the problem
if an neutral atom has ~equal numbers~ of protons and electrons, how many protons does gold have?
 rootbeer003:
rootbeer003:
79
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
sorry i forgot, i thought i put my answer too lol
 rootbeer003:
rootbeer003:
Join the QuestionCove community and study together with friends!
Sign Up
 rootbeer003:
rootbeer003:
@Vocaloid can u help me with more?
 Vocaloid:
Vocaloid:
i'll try
 rootbeer003:
rootbeer003:
ok :)
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
good
Join the QuestionCove community and study together with friends!
Sign Up
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
you might have swapped the abundances
percent Cl-35 * mass Cl-35 + percent Cl-37 * mass Cl-37
= (0.7578)(34.969) + (0.2422)(36.966) = ?
 rootbeer003:
rootbeer003:
ohh c
 Vocaloid:
Vocaloid:
yes
 rootbeer003:
rootbeer003:
Join the QuestionCove community and study together with friends!
Sign Up
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
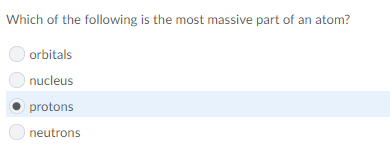
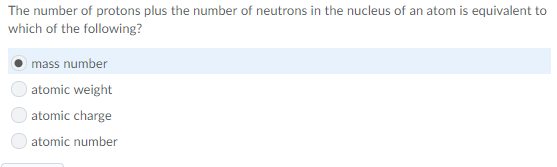
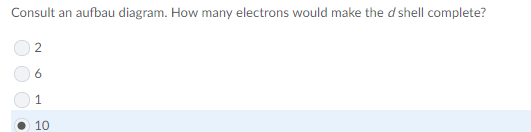
good guess, but since the nucleus includes the protons + neutrons it's bigger than the others combined, making nucleus the better response
 rootbeer003:
rootbeer003:
ohh
 rootbeer003:
rootbeer003:
Join the QuestionCove community and study together with friends!
Sign Up
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
good
Join the QuestionCove community and study together with friends!
Sign Up
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
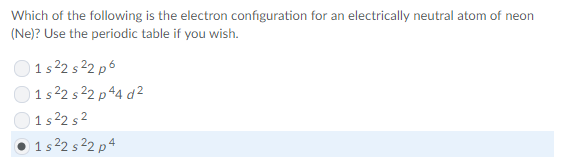
good guess but not quite
since Neon is a noble gas it should have a fully filled p-subshell
based on this, any thoughts?
 rootbeer003:
rootbeer003:
A?
Join the QuestionCove community and study together with friends!
Sign Up
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
 rootbeer003:
rootbeer003:
few more :)
Join the QuestionCove community and study together with friends!
Sign Up
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
 Vocaloid:
Vocaloid:
good
 rootbeer003:
rootbeer003:
ok thanks for helping meee :))