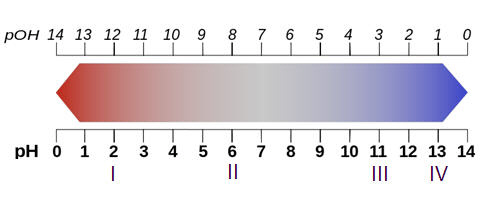
Four different solutions (I, II, III, and IV) are labeled on the pH scale below. Why would solution IV be considered the most concentrated base out of the four substances? -It has the highest concentration of hydrogen ions. -It has the highest concentration of hydronium ions. -It has the highest concentration of oxide ions. -It has the highest concentration of hydroxide ions.
It isn't option A or Option C, which of the last 2 choices do you believe to be correct?
uhh b I think
First of all, the pH is the measure of the acidity of a solution, also pH is the logarithm of the inverse of the concentration of hydronium ions. Therefore, The higher the concentration of hydronium ions the lower the pH. A low pH indicates a high acidity. A high pH indicates a low acidity So, a more concentrated acid is more acid and its pH is lower.
oh okay
um is typing.....
Not sure if that made sense, but, hopefully that helps.
\(\color{#0cbb34}{\text{Originally Posted by}}\) @sealake101 uhh b I think \(\color{#0cbb34}{\text{End of Quote}}\) Not quite
\(\color{#0cbb34}{\text{Originally Posted by}}\) @GalaxyzStarz um is typing..... \(\color{#0cbb34}{\text{End of Quote}}\) hm is typing.....
so it has to be d because it isn't a,b,or c
Yes!
ok thank you @XioGonz and @umm
Anytime. (:
umm is smart
\(\color{#0cbb34}{\text{Originally Posted by}}\) @nalaynana umm is smart \(\color{#0cbb34}{\text{End of Quote}}\) mhm
Join our real-time social learning platform and learn together with your friends!