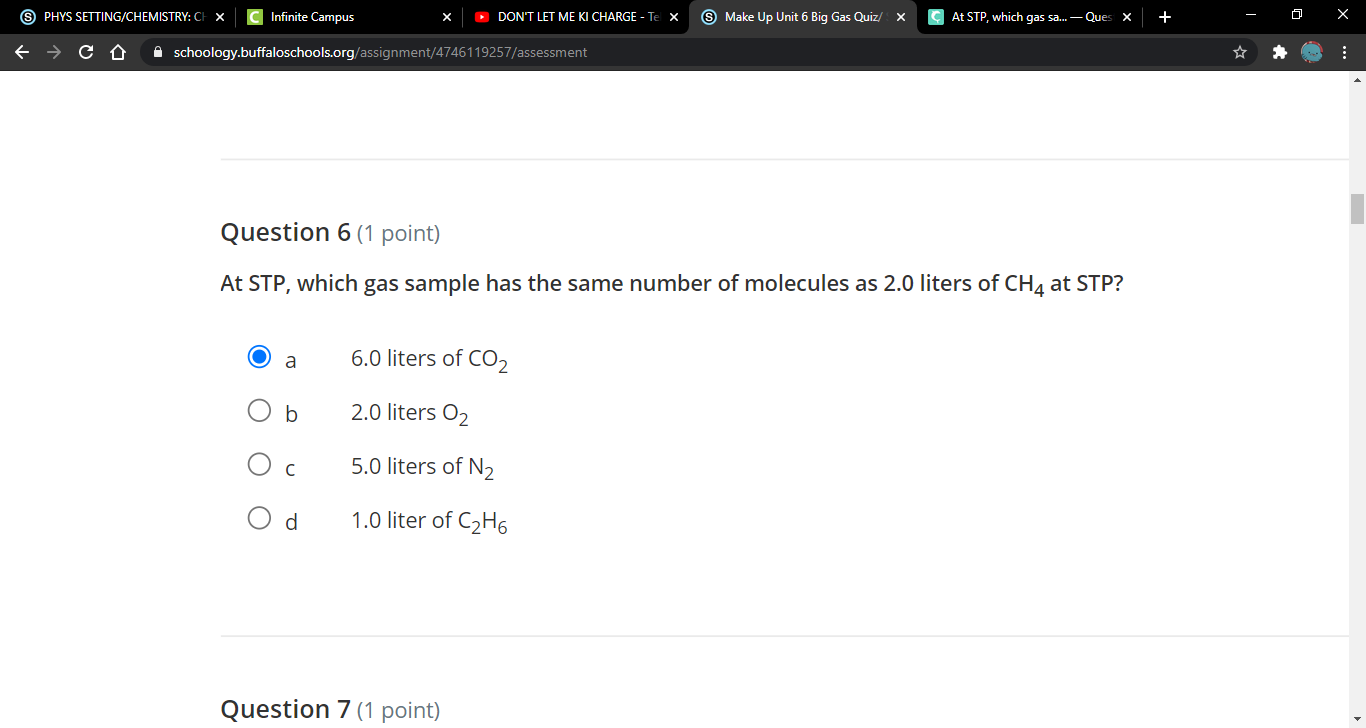
At STP, which gas sample has the same number of molecules as 2.0 liters of CH4 at STP? a 6.0 liters of CO2 b 2.0 liters O2 c 5.0 liters of N2 d 1.0 liter of C2H6
@jhonyy9 can u help with this
ss ?
unfortunately, there's no picture, just the choices
ooh ok i think ik how to do this i thought it was math first
what do you think it is ? just take a guess
I have the feeling it's A
(2) 2.0 L of CH4(g) is your answer. According to Avogadro's law, all gases have equal volume if there are an equal number of particles under the same conditions. there ya go!
\(\color{#0cbb34}{\text{Originally Posted by}}\) @Cuddlybunny (2) 2.0 L of CH4(g) is your answer. According to Avogadro's law, all gases have equal volume if there are an equal number of particles under the same conditions. there ya go! \(\color{#0cbb34}{\text{End of Quote}}\) how is that my answer when that wasn't one of the choices
tsk tsk tsk
\(\color{#0cbb34}{\text{Originally Posted by}}\) @KiWiPoP1 \(\color{#0cbb34}{\text{Originally Posted by}}\) @Cuddlybunny (2) 2.0 L of CH4(g) is your answer. According to Avogadro's law, all gases have equal volume if there are an equal number of particles under the same conditions. there ya go! \(\color{#0cbb34}{\text{End of Quote}}\) how is that my answer when that wasn't one of the choices \(\color{#0cbb34}{\text{End of Quote}}\) XDDDDD
Yea i think its A. tho but idk if its 100% right Just choose whatever best if no one can help
lmao okay, let me shut up b4 I get kicked bih dihopivcdoihvediur you made me hurt my finger again Imma sue you I hope you get the answer wrong-
\(\color{#0cbb34}{\text{Originally Posted by}}\) @Cuddlybunny lmao okay, let me shut up b4 I get kicked bih dihopivcdoihvediur you made me hurt my finger again Imma sue you I hope you get the answer wrong- \(\color{#0cbb34}{\text{End of Quote}}\) smh
Join our real-time social learning platform and learn together with your friends!