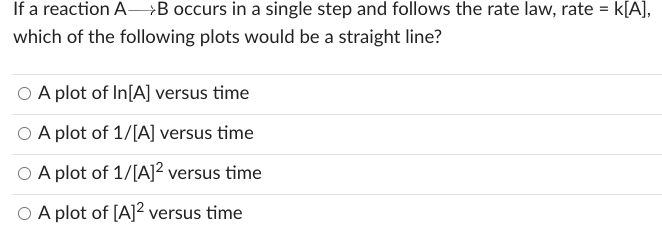Can I get some Chemistry help?
@AZ
@AZ
If a reaction follows the rate constant rate = k[A], what would be the units on the rate constant? Group of answer choices L/mol-s s 1/s mol/L-s
If a reaction ALaTeX: \longrightarrow⟶B occurs in a single step and follows the rate law, rate = k[A], which of the following plots would be a straight line? Group of answer choices A plot of ln[A] versus time A plot of 1/[A] versus time A plot of 1/[A]2 versus time A plot of [A]2 versus time
That's the second question in picture form
one question at a time haha
Okay my bad, thats just two questions and I had to send a picture of the second one because it messed up in the text.
For the first question, it's something you need to understand and then figure it out or you can just straight up memorize it reaction rate has units of M/s but the units for the constant, k, will depend on the order of the reaction so if was a zero order rate = k * [A]^0 rate = k so k has the same units as the rate so it would be M/s (that's Molar which is moles/liter, just in case you weren't aware) if it was first order, now you have rate = k * [A]^1 we know the units for rate are M/s and [A] has units of M so the units for K has to be just 1/s and similarly for second order rate = k * [A]^2 M/s = ?? * M^2 and if you divide M^2 on both sides to determine the units of k in a second order reaction, you'll see that the units are 1/(M*s) so it's just something you need to memorize or if you understand it, something you can just derive and figure out
remember that M = moles of liter so you can replace that but be careful when dealing with the fractions
So the first question would be L/mol-s
I know its not mol/L-s for a fact.
\(\color{#0cbb34}{\text{Originally Posted by}}\) @kamachavis So the first question would be L/mol-s \(\color{#0cbb34}{\text{End of Quote}}\) That's correct
M^-1 s^-1 and 1/M is going to be 1/(moles/liter) = liters/mole
so good job :)
I have a feeling that the second question is "A plot of 1/[A]^2 versus time"
But I am not sure that is why I came here to check behind myself.
\(\color{#0cbb34}{\text{Originally Posted by}}\) @AZ so good job :) \(\color{#0cbb34}{\text{End of Quote}}\) Thank you!
For your second question, once again, you're going to have to memorize the equations and then think of the equations as if they're in the form of y = mx + b t is the variable that changes (basically our 'x') so the slope is going to be k or -k the other term being added is the constant and y would be whatever is on the other side of the equal sign and so the graph would be y vs x
so it would be ln[A]
a plot of ln[a] versus time
\(\color{#0cbb34}{\text{Originally Posted by}}\) @kamachavis a plot of ln[a] versus time \(\color{#0cbb34}{\text{End of Quote}}\) exactly, because it's a first order reaction
Thank you!
You're welcome!
Join our real-time social learning platform and learn together with your friends!