Why is the “kinetic theory of gasses” is important even though it deals with ideal gases not real ones? a Ideal gases and real gases behave exactly the same, making the theory true for both b Real gases deviate slightly from the ideal ones making the theory applicable c Ideal gases are the general case, not real gases, so we don’t have to worry about real gasses' behavior. d We usually deal with gases at high temperature. At very high temperatures, real gases and ideal gases behave the same.
@MrMudd183
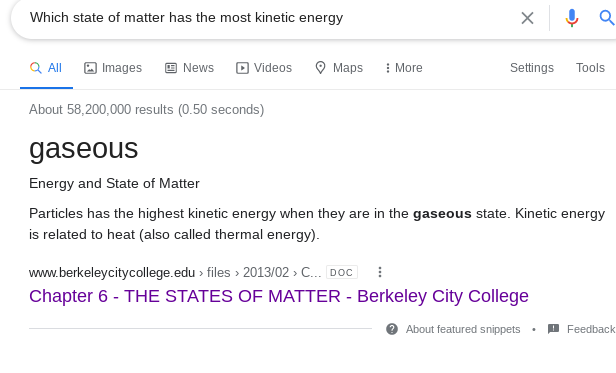
Use this for your notes. http://www.berkeleycitycollege.edu/wp/somar/files/2013/02/Chapter6_The-States-of-Matter.doc#:~:text=Energy%20and%20State%20of%20Matter&text=Particles%20has%20the%20highest%20kinetic,(also%20called%20thermal%20energy).
this link says the anser?
it closes hen i open it
help
Do this,
It's not the answer, but the notes I used in this unit last year.
but i dont understand the page
d?
I woud recomend either answer choice D or andwer choice B. It is your choice, but I am leaning towards D.
Join our real-time social learning platform and learn together with your friends!