Which of the following elements has the highest atomic mass? (3 points) Helium: 2 protons and 2 neutrons Nitrogen: 7 protons and 7 neutrons Oxygen: 8 protons and 8 neutrons Hydrogen: 1 proton and no neutrons
i think its oxygen
It's oxygen. Atomic mass is regularly calculated using the number of protons. Neutrons have nothing to do with this, since it's atomic mass. It has a low combustion, which could be used to trick anyone. Combustion and atomic mass are different. Since oxygen has the highest number of protons, that's your answer.
\(\color{#0cbb34}{\text{Originally Posted by}}\) @XLynx It's oxygen. Atomic mass is regularly calculated using the number of protons. Neutrons have nothing to do with this, since it's atomic mass. It has a low combustion, which could be used to trick anyone. Combustion and atomic mass are different. Since oxygen has the highest number of protons, that's your answer. \(\color{#0cbb34}{\text{End of Quote}}\) This is wrong.
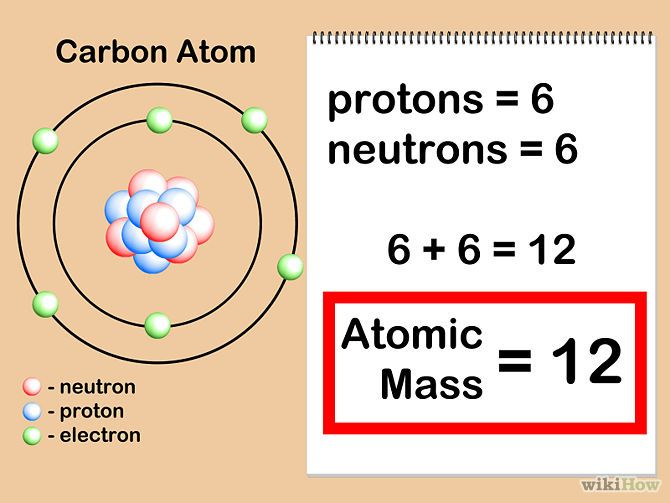
Atomic mass is the mass of 1 atom of that element and that includes it's protons, neutrons and electrons. Remember however, Neutrons and protons mass approximately the same at 1 amu. Electrons are very tiny so it's mass is negligible So when you're trying to find the element with the HIGHEST atomic mass add the number of neutrons and protons whichever one gives you the larger number, that's the element that has a higher atomic mass
So for example, a carbon atom has 6 protons and 6 neutrons (it also has 6 electrons but remember that the weight of an electron is too tiny that we don't even consider it) the atomic mass is going to just be 6 + 6
Obviously, the actual atomic mass is found by looking at the different isotopes of the elements and their frequency but don't worry about that right now the basic basic concept is- atomic mass is made up of the weight of the protons AND neutrons
Join our real-time social learning platform and learn together with your friends!