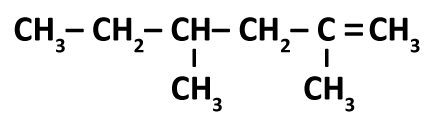
Name the following compound: (Screen shot in comments)
ANSWER CHOICES *2,4-methyl-1-hexane *2,4-dimethyl-1-hexyne *2,4-dimethyl-1-hexene *4-ethyl-2-methyl-1-pentene
Do you know the rules for naming organic compounds? First find the longest carbon chain that contains the C=C You start numbering so that way the the C=C has the lowest numbers If all the carbons are singled bonded to each other, the name would end in -ane If there's one double bonded Carbon, then the name would end in -ene If there's a triple bonded carbon, then the name would end in -yne How long is the carbon chain? If there's only two carbons, then it's ethane three would make it propane four would make it butane five would be pentane six would be hexane and obviously the ane becomes ene or yne depending on if there's a double bonded or triple bonded carbon in the chain
Does that help you reach your answer?
Join our real-time social learning platform and learn together with your friends!