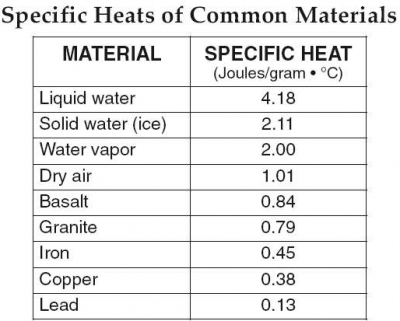
Water has a ______ specific heat. A) insignificant B) low C) high
which one u think it i s?
hope this helps :)
@KiWiPoP1 can you help plz? these ppl aren't being a help at all ;-;
opposite of getting low?
it's c
Ok so your answer isn't gonna be low, cause water most definitely doesn't boil at a low temp, and the rate at which water boils will always be the same, so there's no way it would be insignificant. Water boils at 100 degrees Celsius, so based on this, the answer should be more clear
Ohh okay tyty :)
ywywyw :)
While ultimately you did arrive to the correct answer, I do want to point out something specific heat is completely different than the boiling point specific heat is the amount of heat it takes to raise the temperature of a unit mass by 1 C
Oh yeah, I kinda switched that up
Water has a specific heat of 4.186 J/(g * °C)
Thank you @AZ
And you can compare it to a lot of other materials and water has a very high specific heat relative to other things
Join our real-time social learning platform and learn together with your friends!