Help please: https://snipboard.io/HBQdDE.jpg
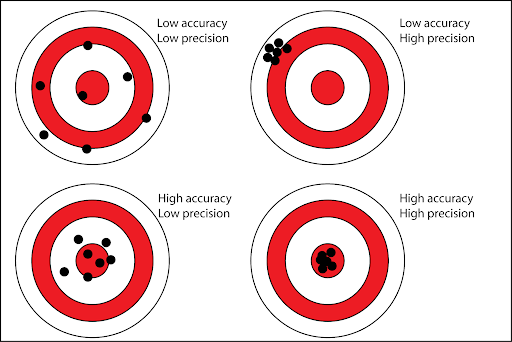
Notice how group A’s average is much closer to the actual value, but the three measurements are all very far apart from each other Group B’s average is very far from the real value, but the three measurements are much closer So, considering the difference between accuracy and precision, what do you think the solution might be?
Yes i am think that Group A is precise and Group B is accurate, but i do not know for sure.
Other way around, group A is more accurate (closer to the real value) while B is more precise (measurements are closer together)
Well it could also be B.)
Because it says that a measurement can be both accurate and precise
Accuracy: Closeness to true value Precision: Closeness of a set of data points to one another
So in this case i think the answer might be B.) what do you think?
I don’t agree with B. 13.2g is too far off from 10.5 for group B to be accurate.
Ohhhh we have to use 10.5g/cm^3 sorry i was reading the question all wrong
I agree with option C.) that group A is accurate and Group B is precise
Yup good
Join our real-time social learning platform and learn together with your friends!