Ask
your own question, for FREE!
Chemistry
6 Online
chemistry
Still Need Help?
Join the QuestionCove community and study together with friends!
@vocaloid
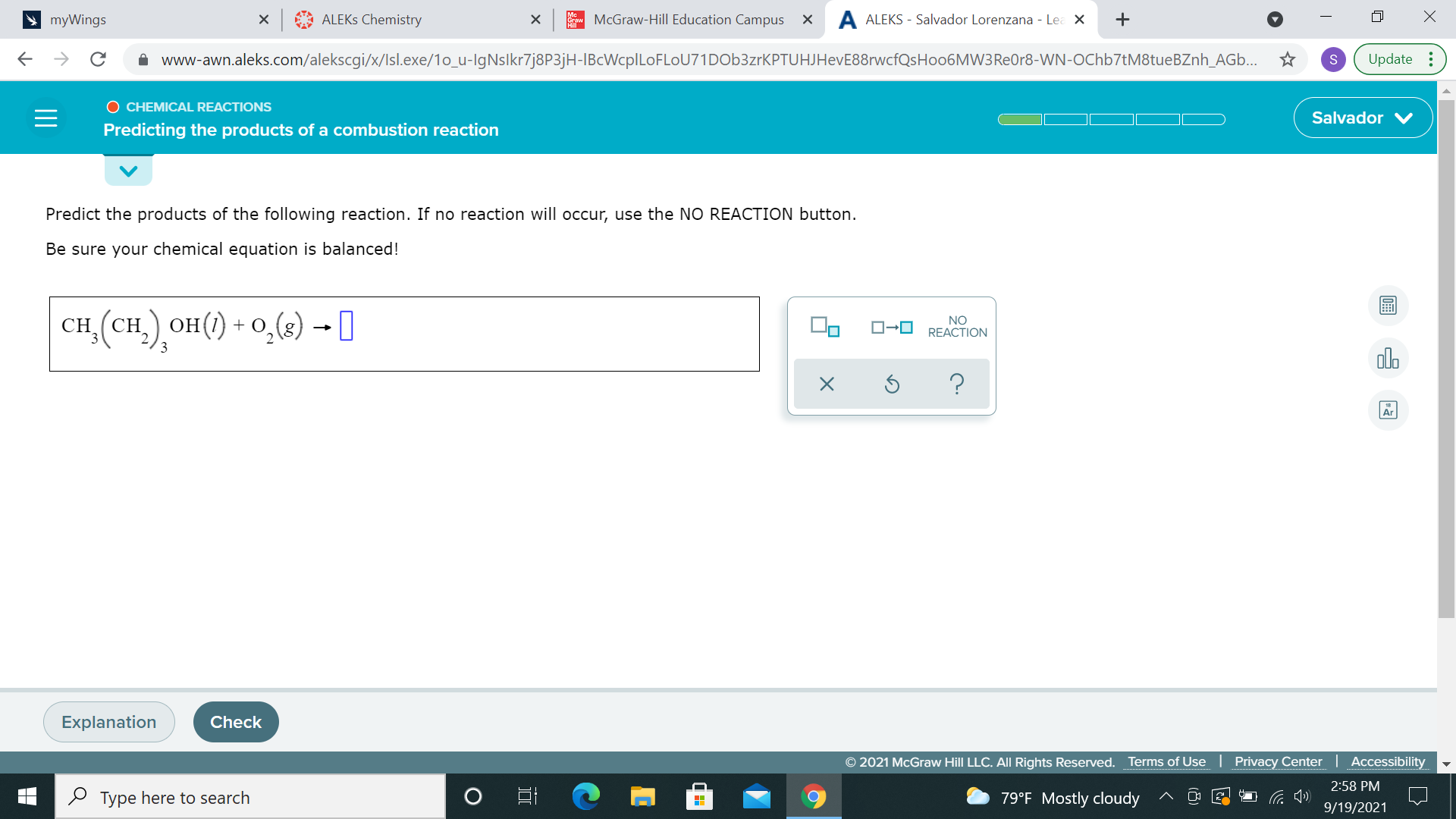
another combustion reaction (left compound is ethanol, which is in gasoline) so your products are CO2(g) and H2O(g) again, balance
im not sure how to balance
put a 5 in front of H2O?
Still Need Help?
Join the QuestionCove community and study together with friends!
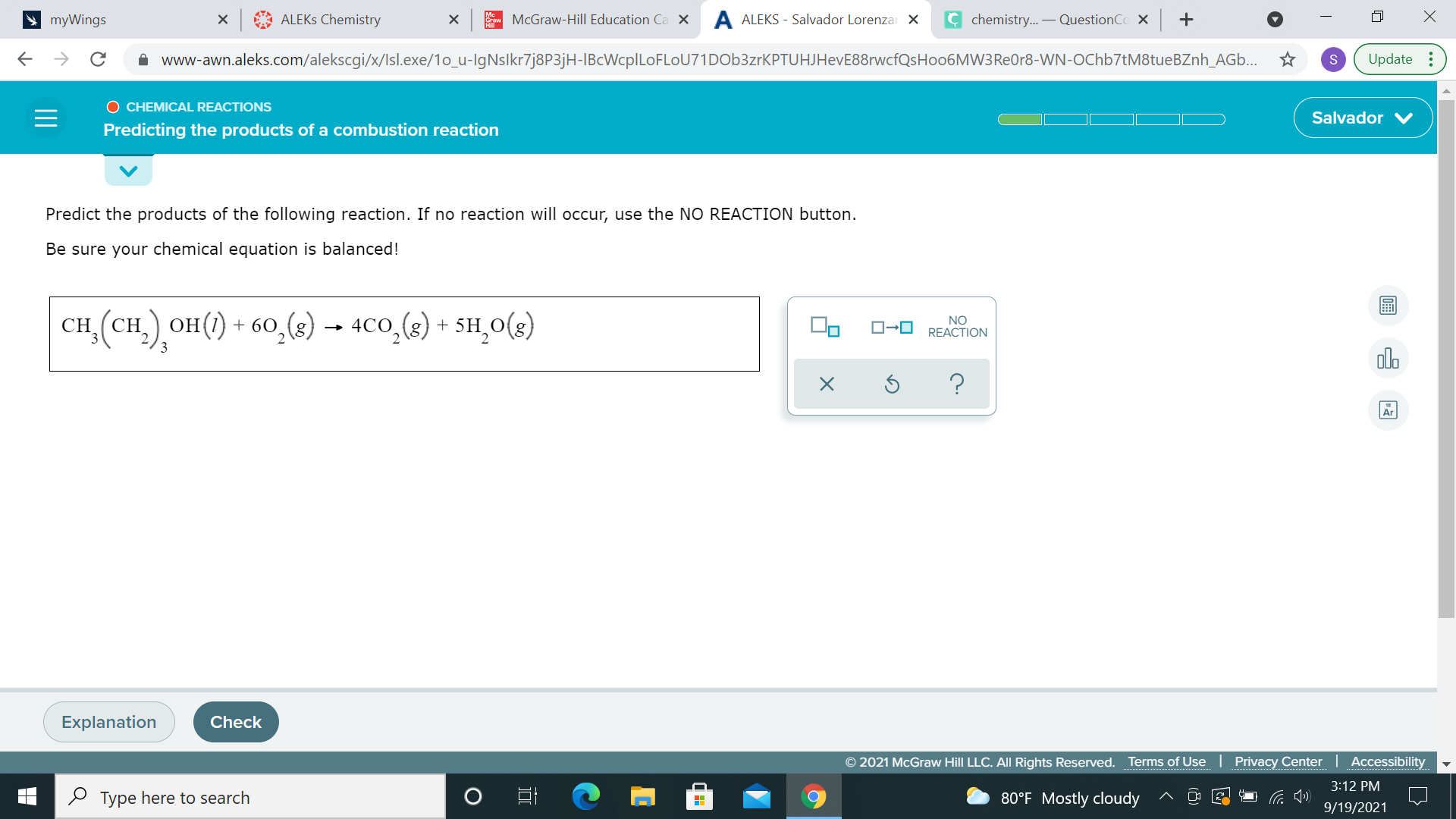
yup, that's the step I would do then I would try balancing out carbon, then oxygen at the end
put a 4 infront of CO2?
yup, then balance oxygen
yup everything looks balanced
Can't find your answer?
Make a FREE account and ask your own questions, OR help others and earn volunteer hours!
Join our real-time social learning platform and learn together with your friends!
Join our real-time social learning platform and learn together with your friends!
Latest Questions
 MrsTooTac:
pedos nowadays ud83dude4fud83cudffeud83eudd26ud83cudffeu2640 . its alot on here i can name .
MrsTooTac:
pedos nowadays ud83dude4fud83cudffeud83eudd26ud83cudffeu2640 . its alot on here i can name .
 Midnight97:
Kinda a roleplay story between me and my friend enjoy... Part one Forgive me for all the screenshots.
Midnight97:
Kinda a roleplay story between me and my friend enjoy... Part one Forgive me for all the screenshots.
 StevenisGhost:
what type of song should I make next, and will y'all go check out my new song on
StevenisGhost:
what type of song should I make next, and will y'all go check out my new song on
 Midnight97:
My drawing sure changed over the years look at these two pictures from 2024 to no
Midnight97:
My drawing sure changed over the years look at these two pictures from 2024 to no
 EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
 EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
 Breathless:
I don't know if this would be considered art, but its close enough I believe, Any
Breathless:
I don't know if this would be considered art, but its close enough I believe, Any
 Demon25:
Let my silence be my voice Let my silence remind you how many times I tried speak
Demon25:
Let my silence be my voice Let my silence remind you how many times I tried speak
15 hours ago
0 Replies
0 Medals
19 hours ago
2 Replies
1 Medal
1 day ago
5 Replies
1 Medal
19 hours ago
6 Replies
1 Medal
3 days ago
3 Replies
0 Medals
5 days ago
0 Replies
0 Medals
1 week ago
3 Replies
0 Medals
1 week ago
5 Replies
1 Medal