VESPR
Why does this make sense? Written out, the Cl is supposed to have 5 electron groups, 4 of which are singular bonds and one is a lone pair. Which makes out as 5 electron group and 1 lone pair equalling out as a see-saw structure. However this homework assignment said it was the answer given above. Am I right and do I have to go hunt down my professor?
What is the molecule?
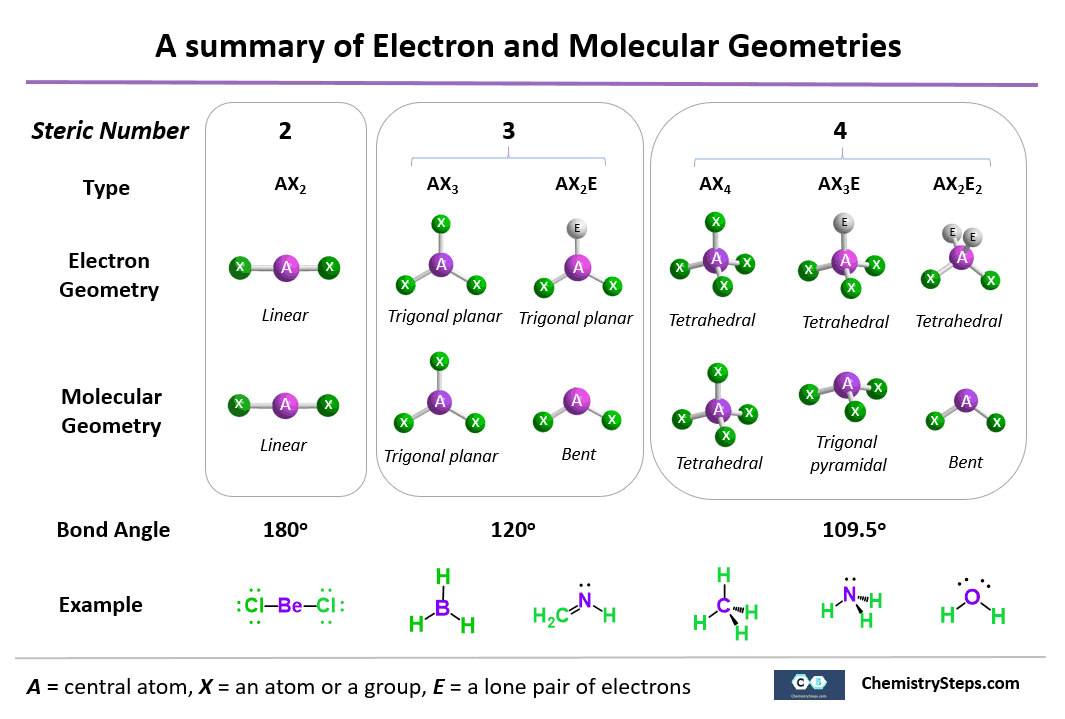
There's a distinction between molecular geometry and electron geometry In electron geometry, it doesn't matter if it's a lone pair of electrons or part of a bond. You consider them the same and so you it would just be trigonal bipyramidal. If they were asking for the molecular geometry, then if there's 5 electron groups and 1 is a lone pair then yes it would be the seesaw structure Here's a nice image that displays this difference between electron and molecular geometry
Join our real-time social learning platform and learn together with your friends!