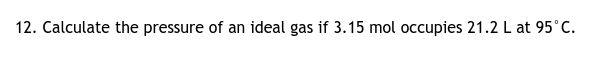Ask
your own question, for FREE!
Chemistry
6 Online
umm help i forget how to do this
Still Need Help?
Join the QuestionCove community and study together with friends!
it's stated to be an ideal gas, so you can use the formula PV = nRT where P is pressure, V is volume, n is moles, R is the ideal gas constant, T is temperature some caveats: 1. you must convert temperature in C to temperature in K by adding 273.15 2. because our units are now L, moles, and K, we can use the gas constant R = 0.0821 L·atm/mol·K. remember that different units have different R values. plug in the appropriate quantities and solve for P, pressure. based on our R value, your answer will be in atm.
ohh okok thank you
Can't find your answer?
Make a FREE account and ask your own questions, OR help others and earn volunteer hours!
Join our real-time social learning platform and learn together with your friends!
Join our real-time social learning platform and learn together with your friends!
Latest Questions
 MrsTooTac:
pedos nowadays ud83dude4fud83cudffeud83eudd26ud83cudffeu2640 . its alot on here i can name .
MrsTooTac:
pedos nowadays ud83dude4fud83cudffeud83eudd26ud83cudffeu2640 . its alot on here i can name .
 Midnight97:
Kinda a roleplay story between me and my friend enjoy... Part one Forgive me for all the screenshots.
Midnight97:
Kinda a roleplay story between me and my friend enjoy... Part one Forgive me for all the screenshots.
 StevenisGhost:
what type of song should I make next, and will y'all go check out my new song on
StevenisGhost:
what type of song should I make next, and will y'all go check out my new song on
 Midnight97:
My drawing sure changed over the years look at these two pictures from 2024 to no
Midnight97:
My drawing sure changed over the years look at these two pictures from 2024 to no
 EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
 EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
EdwinJsHispanic:
"poem" love is So Beautiful to have. But it's so hard to have. At this point I don't know whether its worth the wait Or if it's just millions of miles to re
 Breathless:
I don't know if this would be considered art, but its close enough I believe, Any
Breathless:
I don't know if this would be considered art, but its close enough I believe, Any
 Demon25:
Let my silence be my voice Let my silence remind you how many times I tried speak
Demon25:
Let my silence be my voice Let my silence remind you how many times I tried speak
16 hours ago
0 Replies
0 Medals
21 hours ago
2 Replies
1 Medal
1 day ago
5 Replies
1 Medal
21 hours ago
6 Replies
1 Medal
3 days ago
3 Replies
0 Medals
5 days ago
0 Replies
0 Medals
1 week ago
3 Replies
0 Medals
1 week ago
5 Replies
1 Medal