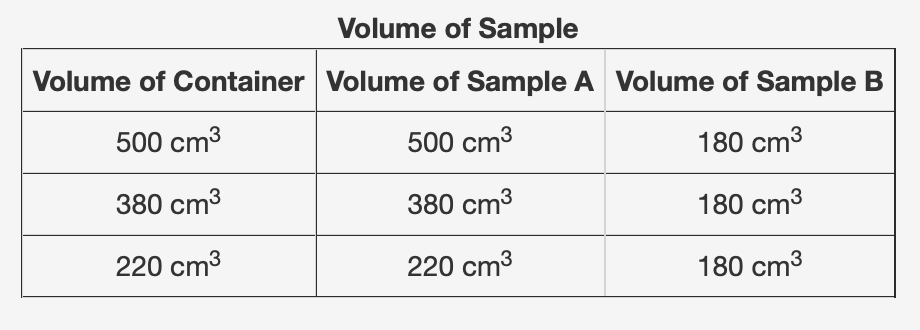
The table below shows the volume of two samples, A and B, when placed in three containers of different volumes. Which of the following correctly describes the state of matter of one of the samples? A. A is a solid because its volume is equal to the volume of the containers. B. B is a gas because its volume is less than the volume of the containers. C. B is a solid because it contracts as the containers change. D. A is a gas because it contracts as the containers change.
the table:
@snowflake0531
ok, so what do you already know?
honestly, I dont know. I forgot-
well i cant help sorry :(
wait what?
why cant you help :')
@tranquility
i also getting confused
i am *
So what would you guess/think it is?
tbh I think its D but im not sure
No, D would be incorrect.
oh, how so?
seems like he doesn't want to respond
You have solids, liquids and gases solids will maintain the same volume and the same shape liquids will maintain the same volume but they will take the shape of their container Just think of different shaped classes and you're transferring the same amount of liquid from one glass to the next. The volume stays the same but the shape will change depending on if you're pouring it into a bowl or a vase. A gas does not have a fixed shape or volume. Gas will take the shape of the container and you can compress it so it's volume can be decreased to fit the container. Obviously there's a limit and you can't compress it forever, but until a certain point. The pressure would be building up as you compress it into a smaller container.
Join our real-time social learning platform and learn together with your friends!