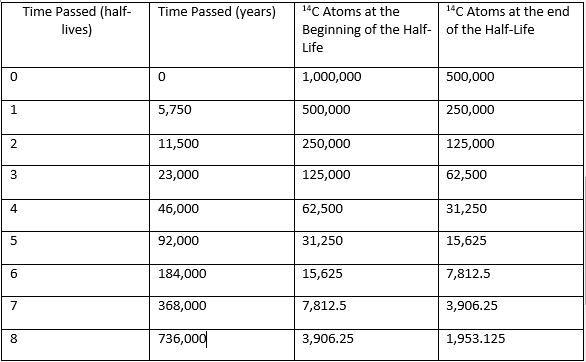
Scientists have determined that the half-life of 14C is 5,750 years. Use that information or complete the table, which begins with 1,000,000 atoms of 14C Table: http://i.imgur.com/HqwgkM2.jpg
A half life is the time it takes a substance to decrease by half.
I know that, but am I just supposed to decrease the number of atoms by half each half-life passes and double the amount of years that went by? or something else entirely? XD
Yes. See in the years spot, it shows you the full life of the atom. In the time passed and the years, they are zero, so at the beginning of half life, the atom is at it's fullest. In the end of the half life, just subtract the full term by half.
Okay, thank you so much!!
Your welcome.
would this be right?
Yes it looks fine.
Awesome, thank you again for all your help! It's grately appreciated!!!
Anytime.
Join our real-time social learning platform and learn together with your friends!