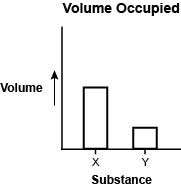
The graph below compares the volume occupied by 100 g of liquid water and 100 g of water vapor. Which statement best describes the two substances? A. Particles are farther apart in liquids so substance Y is liquid water. B. Particles are closer together in liquids so substance X is liquid water. C. Particles are closer together in gases so substance Y is water vapor. D. Particles are farther apart in gases so substance X is water vapor.
I have the chart right here but i know its not a or c. @Allison @Ultrilliam
Looking at the graph, the Y axis is volume occupied, so the higher the representation of the substance, the more volume it occupies. Now, water vapor is water in a gaseous state and gases have more space spread apart between them and are in smaller particles, because of this, the gas is able to occupy any amount of space that the container allows. In other words, the bigger the container, the more volume the gas is occupying.
Your a life saver, thank you so much!! P.S i am gonna be your fan(:
Happy to help.
Join our real-time social learning platform and learn together with your friends!