Would this be 5.42 * 10^24
@Shadow @Vocaloid
did they give you a molar mass of carbon13?
they gavce me carbon12= 6.02*10^23
huh. strange. molar mass of carbon-13 is 13.00335g/mol according to the internet so you would just divide 9.000 by 13.00335 to get moles i think
i got 117
but its wrong when i punch it in
divide not multiply
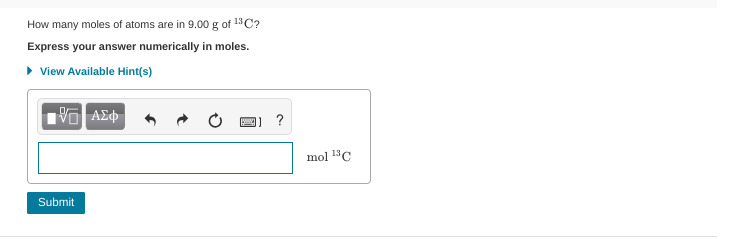
9.000/13.00335
and yes I went in the hints and the mass is 13 so I did 1*9.00
13**
.621?
.692? ***
.692 should be it
Based on your answer in Part A, calculate the number of atoms in this amount of 13C?
oh to go from moles to atoms just multiply the # of moles by 6.022e23
7.82 * `10^24
7.82? we got 0.692 as the # of moles, right? so .692 * 6.022e23
oh okay I was doing 13 ... 4.17 * 10^23
good
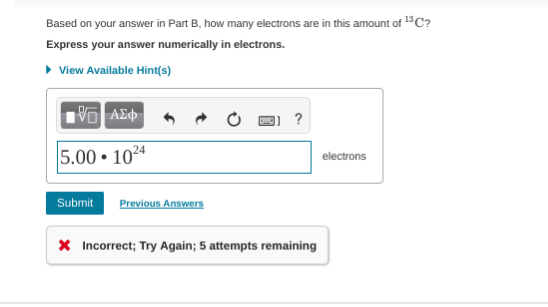
Based on your answer in Part B, how many electrons are in this amount of 13C?
carbon 13 has 12 electrons so multiply the # of molecules by 12
156
4.17 * 10^23 is the number of molecules so 12 * 4.17 * 10^23
5.0 * 10^24
5.00 * 10^24 since we have three sig figs
oh wait
carbon-13 has 6 electrons not 12 >> so 6 * 4.17 * 10^23
2.50*10^24
good, lmk how that turns out
Based on your answer in Part B, how many neutrons are in this amount of 13C?,, 13 for this
each 13C atom has 7 neutrons 7 * 4.17 * 10^23
D?
hm, not quite, try going back to A or B and rechecking the calculation
b
good
.26?
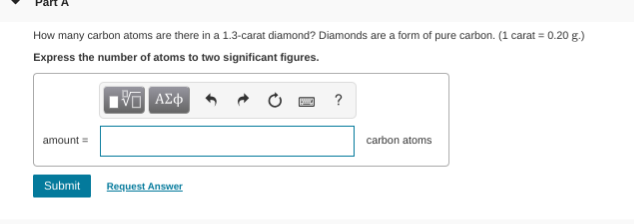
they give you 1 carat = 0.20g therefore 1.3 carat = 0.20 * 1.3grams going from grams to moles, divide by the molar mass of carbon (which is about 12.011g/mol) then multiply avogadro's # (6.022e23) to get atoms
hm I got something a little diff. remember, 1 atom is tiny so you would expect a lot of atoms
.26*12.011 * 6.022*10^23=== 1.88*10^24
very close. after you get 0.26g, you need to divide by 12.011 not multiply so (0.26/12.011)*6.022e23 should do it
1.30*10^22
only 2 sig figs so 1.3 * 10^22 not 1.30
Calculate the mass of 2.25×1022 tungsten atoms. I would utilize do (2.25*10^22)*(6.022*10^23)
hm, close, since you have atoms and you want to go to moles, you would first divide by avogadro's number, then multiply by the molar mass
|dw:1537325793408:dw|
what would the atoms be?
so you are given 2.25×1022 tungsten atoms. according to the chart, divide by avogadro's number 6.022e23, and then from there, the chart says to multiply by the molar mass of Tungsten (get this from the ptable)
6.87
good
D
perfect
Join our real-time social learning platform and learn together with your friends!