Help with this. When you get a chance
|dw:1538520412181:dw|
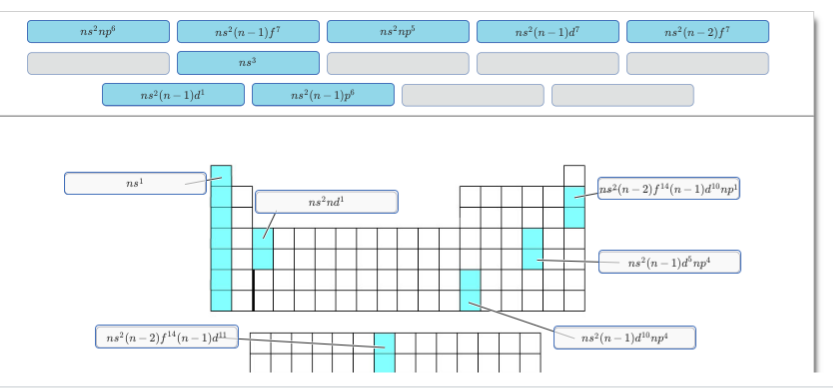
upper right corner: should be s and p block only, notice how Argon stops before the dblock if you are having trouble you can think about the two elements that actually have that configuration, Neon and Argon [He] 2s2 2p6 [Ne] 3s2 3p6 see if you can figure out which choice accurately describes both configurations
for the next one below it, you almost have it, but the d needs to have 10 electrons since both those elements have a full d block (notice how you have to go all the way across the d block to get those two elements)
for the one below that you have a fully filled f-block so the one with f14 is your only possibility
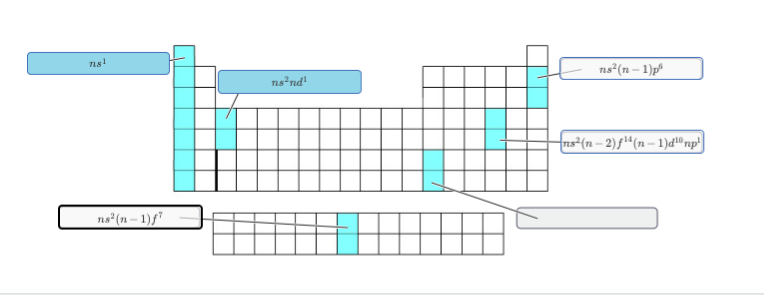
finally for the lower left corner you only have 7 electrons in the f block so you only have two chocies ns2(n-1)f7 or ns2(n-2)f7 looking at the actual electron configurations for Europium and Americium [Xe] 4f7 6s2 [Rn] 5f7 7s2 should make it clear whether it's n-1 or n -2 for the f block
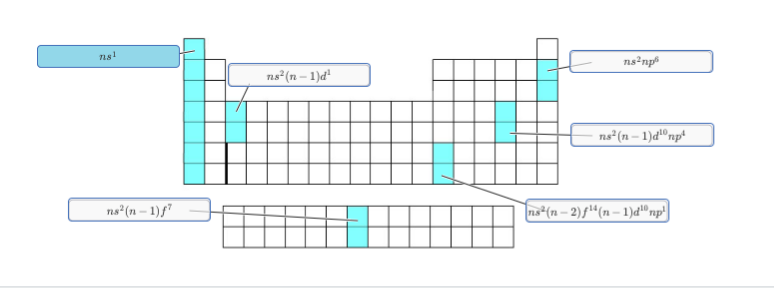
upper right corner: [He] 2s2 2p6 [Ne] 3s2 3p6 since n is the same for the p block and s block it should just be ns2 np6
for the one below it you originally had ns2(n-1)d5np4 you just needed to fill up the d block to get ns2(n-1)d10np4 the f-block is not involved
for the third one on the right (where thallium would be) you have a full f-block so ns2(n-2)f14(n-1)d10np1 is your only possibility
also I didn't really catch this before but the second one (where scandium and yttrium would be) it needs to be ns2(n-1)d1 because the outer configuration is 4s2 3d1 and 5s2 4d1
almost notice how the bottom configurations for americium and europium are [Xe] 4f7 6s2 [Rn] 5f7 7s2 so it has to be n - 2 since 4 is 2 less than 6; 5 is 2 less than 7
otherwise it's good
okay so n-2 instead of n-1
yes
Join our real-time social learning platform and learn together with your friends!