Okay so I am just having trouble with what element this would be?
|dw:1538532034499:dw| should be ar-senic (this website will filter it out if you don't put the dash) it has a full d-block see what you can get from this
Could you explain this to me I don't get it. Like how I would find the element I know how to do electron configuration though
start from hydrogen (row 1) go down to the 4th row (starts with potassium K) group 5A is the 5th column (ignoring the transition metals) which is the column starting with nitrogen
this is kind of confusing but there are two numbering systems for groups |dw:1538532364152:dw|
C?
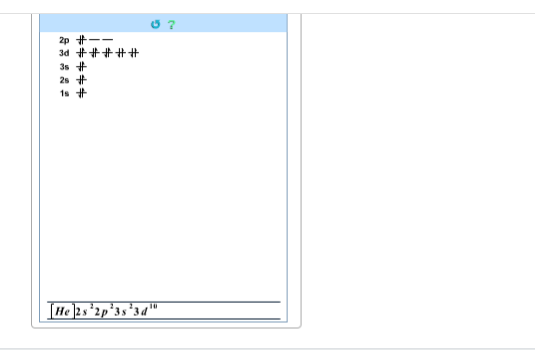
Okay so I just googled it and the answer is a .... But, how is it the d value.
notice how Ars-enic comes after a full d block so you would expect d10
Isnt there an empty space going across the d block
but its empty... does it still count as d10
even if there aren't any elements
empty? the electrons from Sc to Zn are full
|dw:1538532708081:dw|
Oh okay I see.
yeah that should be it the only other possibility would be 7A but let's try 17 for now
It worked
Sodium
that's a good guess but sodium is considered an alkali metal |dw:1538533859032:dw| the only nonmetal in the list is phosphorus
anything above the zigzag?
yes
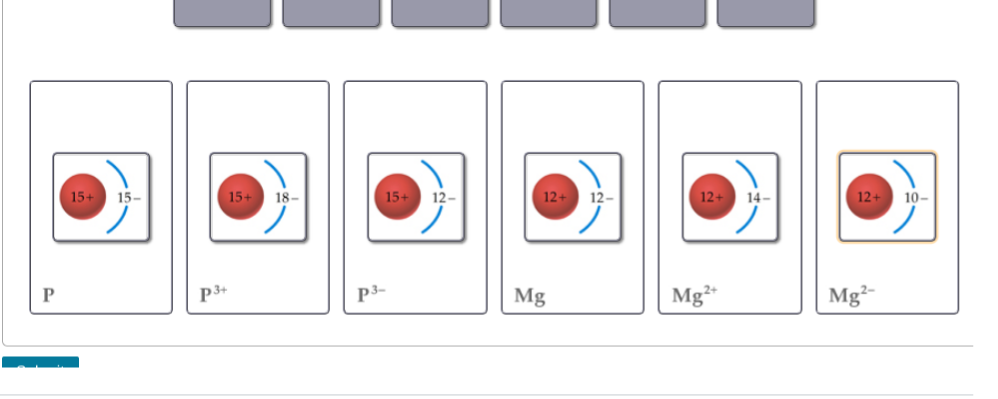
Did I match it correctly
you need to switch P3+ and P3- and Mg2+ and Mg2- if there are more electrons than protons the overall charge will be negative
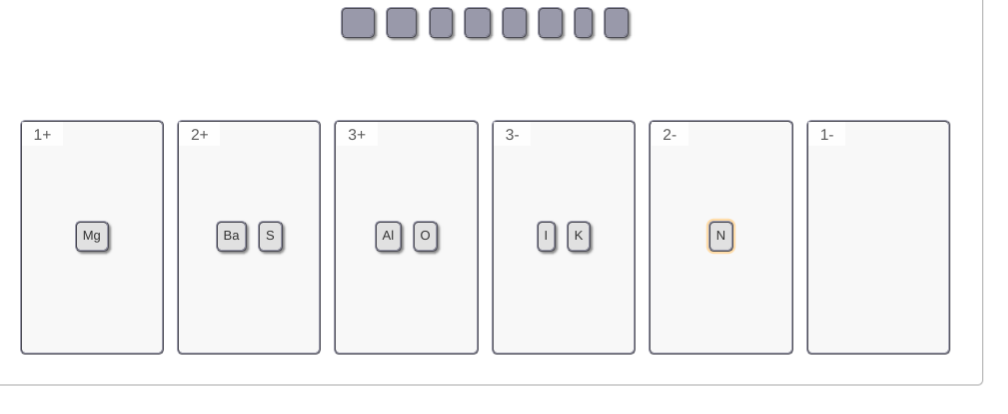
pay attention to the group # and position on the ptable. elements toward the left side generally make positive ions and elements towards the right side generally make negative ions K is in group 1 so it gets a +1 charge Mg and Ba are in group 2 so they get a 2+ charge S and O are in group 6A so they need 2 electrons to get the full 8 valance electrons, so they'll take 2 extra electrons to get -2 charge Al is always +3 (you just need to memorize this) N needs 3 electrons --> -3 charge I only needs 1 electron --> -1 charge
In the ground-state electron configuration of Fe3+, how many unpaired electrons are present?
4?
I got [Ar]4s^0 2d^5
so would it be 4 or 5 because the 4s there isn't any paired and then for 2d5 there is one that is unpaired?
each of the 5 electrons in the 2d subshell gets its own orbital, so 5 unpaired
oh right. but what about the 4s^0
would they still be paired even with the zero
4s^0 means no electrons are in the s orbital so they're not available for pairing
oh got it
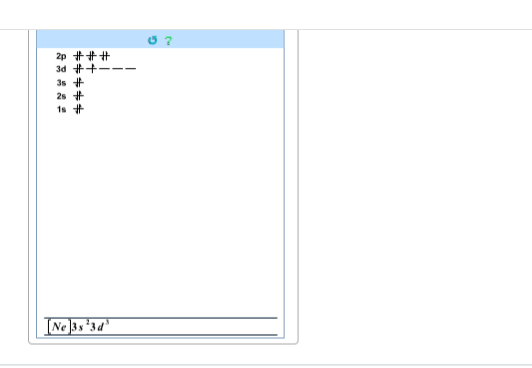
Build the orbital diagram for the ion most likely formed by phosphorus.
I drew this
hm not quite Phosphorus wants to attain the same electron config. as the next highest noble gas which is Argon going from Neon to Argon we have [Ne] 3s2 3p6
oh okay so the answer is [Ne]3s2 3p6
But wouldnt it be the noble gas before that?
like the one before p is Ne
right, so the previous noble gas is Ne so we'd use Ne instead of helium
oh right
I just inputted it wrong. I did orginally do 2p but i guess it didnt show up
the d-block should be empty as well
but it says an ion formed by phosphorous it would be the same right
so no 3d
right
it should be the same configuration as Argon
[Ar]3d^10
because it would condense down to Ar
it still needs to start with Neon
idk why its condensing down
so phosphorus wants to match the higher noble gas (Argon) the configuration of Argon is 1s2 2s2 2p6 3s2 3p6 which is equivalent to [Ne] 3s2 3p6
Oh 3p^6 :S
why is the 2p orbital above the others it should be between 2s and 3s
I got [Ne] 3s2 3p6 now
Is that right
the configuration is right, idk what's going on in your orbital diagram
Fixed it
Ugh it says its wrong:/
you need to switch 3s and 2p on the orbital diagram if the configuration itself is wrong idk
wait so which comes first
2p?
1s2 2s2 2p6 3s2 3p6 2p comes before 3s
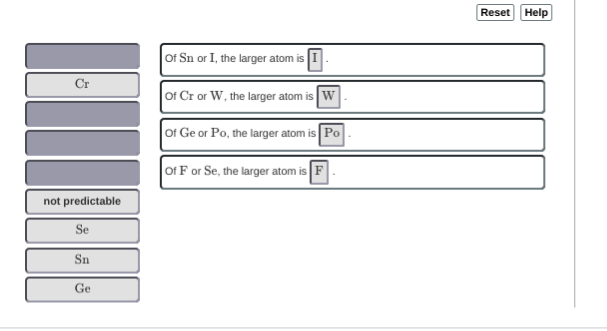
iirc phosphorus should be larger than sulfur since it's closer to the left side of the ptable otherwise i think it's good
almost, for the last one Se is the bigger one (further down the group and further left on the ptable) Fluorine will pretty much be smaller than everything
it says its wrong
You filled in 2 of 4 blanks incorrectly. Note that Sn and I both reside in period 5 of the periodic table (belonging to different groups). Consider the trend of atomic radius as you move right across a period.
this is what it says
huh
it syas its wrong and then gave me that^ explanation
oh duh, the further right you go the smaller the atoms are >_> so Sn will be greater than I
and then cr is greater than w?
hm, no, W should be bigger
okay then one more that they are saying is wrong
oh ge?
is bigger?
just a hunch but they might want "not predictable" for Ge vs Po :S
okay so we got Sn , W, np, and f
it says one blank is wrong
Se is bigger than F
\(\color{#0cbb34}{\text{Originally Posted by}}\) @Vocaloid almost, for the last one Se is the bigger one (further down the group and further left on the ptable) Fluorine will pretty much be smaller than everything \(\color{#0cbb34}{\text{End of Quote}}\)
Got that one.. For this this is the radius size
So would it be the same concept
Rb,Ca,,S,Si,F
Thats what I put
Silicon is bigger than sulfur (it's closer to the left side) Rb,Ca,,Si, S,F
C
hm, close but not quite, smaller atoms mean more charge, so Cl should have a larger ENC (effective nuclear charge) than P
a
So then a right
yeah that's what i got too
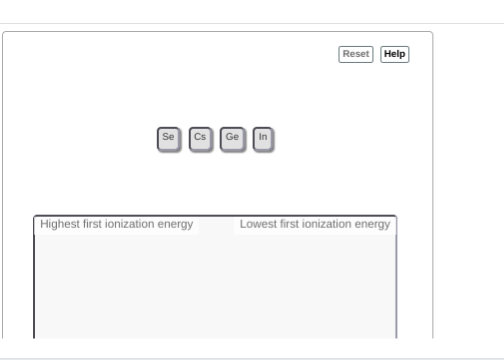
Ge,Cs,In,Se?
|dw:1538538277806:dw| hm not quite, since it wants high to low ionization energy try going from the top right to the bottom left corner of the ptable
,In,Se, Ge, Cs?
almost Se is the most towards the upper right going straight left gives us Ge then In, then Cs at the bottom
yikes you need to draw out the orbital diagrams, and if there are unpaired electrons it's paramagnetic, if all the electrons are paired its diamagnetic
5 mins ahh
uh going from right to left F, O, N, B, Na, Li?
there may be some wonky stuff going on with O and N though
actually wait, switch the N and O, there's a full subshell on N which makes the energy higher
Which of the following trends is indirectly proportional to effective nuclear charge, Zeff?
ionic energy welectron affinity atomic size all the answeres are correct?
atomic radii (just have to memorize this)
or atomic size i gues
Of Ca or Sr, the element with the higher first ionization energy is
Ca?
yes
Join our real-time social learning platform and learn together with your friends!