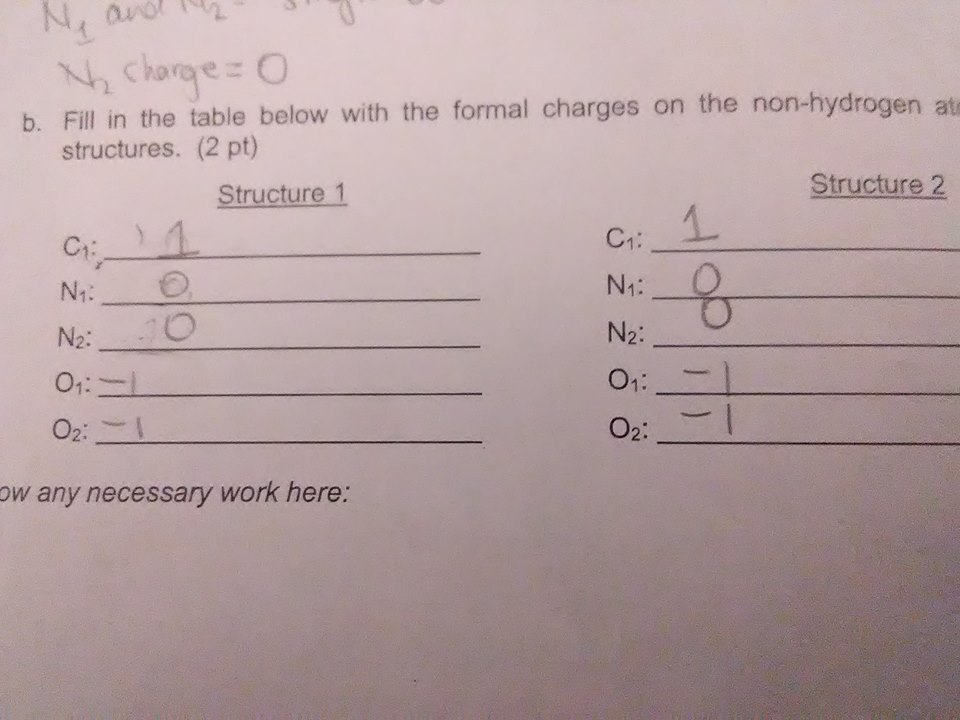WHich ion, chlorate or chlorite would you predict to have a larger O-CI-O bond angle? Briefly justify why
any attempts to draw out the lewis structures/geometries? as a hint chlorate has one more oxygen than chlorite, what would that do to the bond angle?
Would that bring the steric number
Be the **
:S
yeah, if you want to formally calculate the geometries then sure you can calculate the steric number
Okay so it woul be chorite then?
because its steric numner would be 4
hm. i'd have to think a bit more about this.
|dw:1540515268325:dw|
since chlorate is trigonal pyramidal (3 bonds, 1 lone pair) and chlorite is bent (2 bonds 2 lone pairs, chlorite acually has the slightly smaller bond angle contrary to what I expected
it's really close though and could honestly go either way tbh
so chlorate has the larger bond angle then
yeah, those are my thoughts (I should probably double check w/ someone though)
@lowkey
meugh different sources are giving me different values for the bond angles |dw:1540515880044:dw|
|dw:1540515997522:dw| my conclusion is that electron pairs cause more electron repulsion than electronegative atoms, so chlorite with 2 lone pairs and 2 bonds has a smaller angle than chlorate which has 1 lone pair and three bonded atoms
DO you want to see the sheet my professor provided>
sure
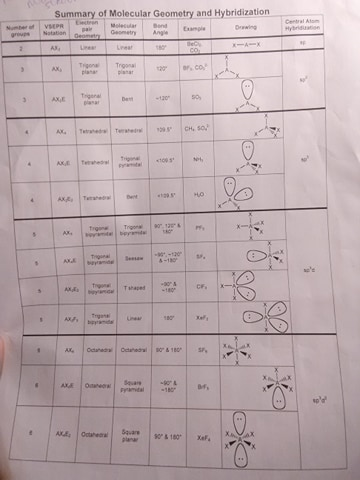
yeah I was thinking it would be like that in this case both of the compounds are classified as <109.5 so your sheet doesn't really distinguish them properly
OKAY SO just stick to the original answer
yeah, I believe chlorate would have the larger bond angle
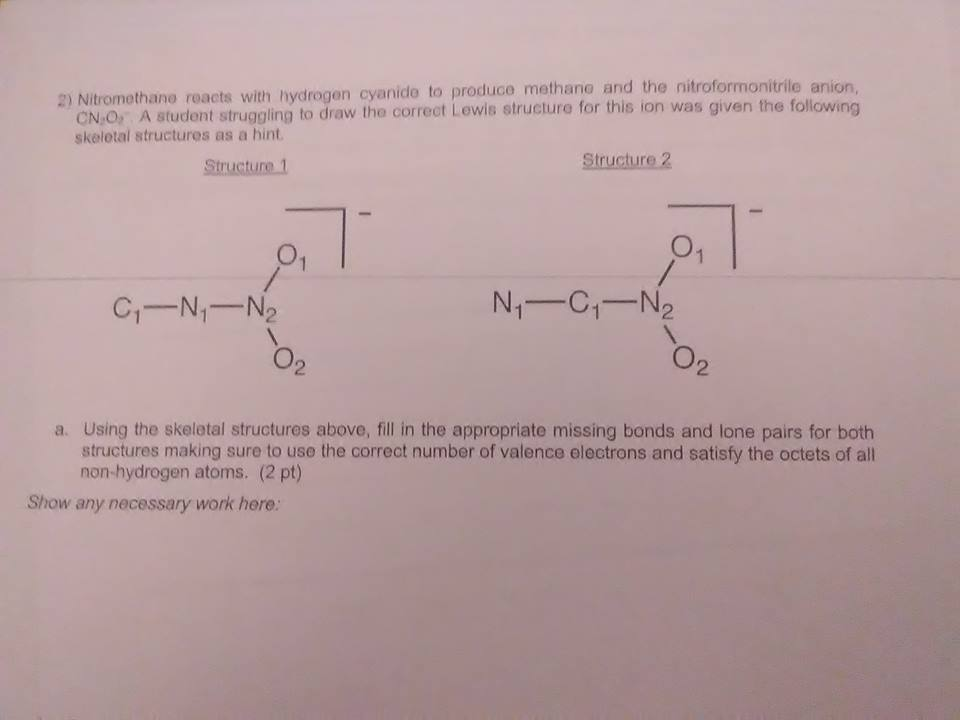
oh well, you just gotta fill in the appropriate # of missing bonds or lone pairs so that each atom has its octet filled
on the actual structure right?
yeah, this might take me a bit tbh
as a hunch the -1 formal charge would end up being on one of the oxygens and the rest would have formal charges of 0
wait so I do it in the a section I'm cofused
and if i do it on the structure where ? :S
I think so, you would just draw extra bonds on the structure and extra lone pairs on the atoms
|dw:1540517477626:dw|
|dw:1540517482384:dw|
something like that
the one one the left is tripping me up b/c the C would almost certainly never be on the left side of this atom
its only gen chem so maybe not thinking about all the rules and just kind odf simplifying it you know
its really just general chemistry nothing advanced
|dw:1540517596682:dw|
I can't get the formal charges to sum up to -1 for some reason >>
Me either
wth
the triple bond ? :S
I just checked total # of bonds and since we have an odd # of electrons there may be a lone electron somewhere
Ont he nitrogen
its octet isn't full yet :S
nvm it is :/
what about on the O1
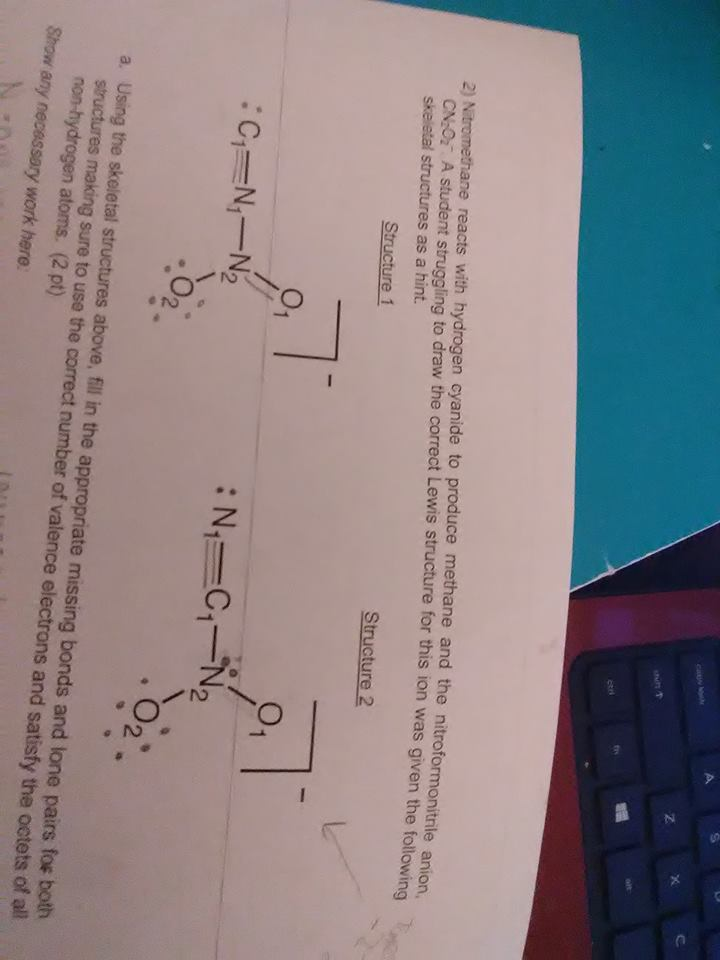
well using process of elimination left structure: the bond between C1 and N1 is probably a triple bond, making the bond between N1 and N2 a single bond giving N2 one lone pair gives N2 a formal charge of 0 then just fill up the electrons on the oxygens giving them both a formal charge of -1 it's not the right formal charge but it's the best I came up with
right structure: give N1 a lone pair and triple bond N1 with C1 single bond C1 with N2, give N2 a lone pair, and single bond N2 with the other oxygens, giving the oxygens a formal charge of -1
then for b you'd just calculate the formal charges then for c) you'd circle the structure that gives EN atoms negative formal charges and electropositive atoms positive formal charges putting carbon on the left side forces it to have a negative formal charge so structure 1 is very unfavorable
idk if it's a typo on the worksheet but after doing the work I have a hunch the overall formal charge was meant to be -2 not -1
So that for the first part
yeah just don't forget the lone pairs on both O1's, you want a full octet
just one lone pair?
so two dots
oxygen requires a full octet so it must have a total of 4 pairs of electrons, either bonded or lone pairs so for O1 you already have 2 bonds, how many lone pairs do you need to fill the octet?
2
2 bonds are 4
good what about the other O1? it only has 1 bond so how many Lone pairs does it still need?
so four more so 2
4
hm, close, the other O1 only needs 3 lone pairs to get a full octet
yeah i miscounted
almost just a few issues Structure 1: C1 has valency of 4; 1 lone pair; 3 bonds --> formal charge = ? N1 has valency 5; 0 lone pairs, 4 bonds ---> formal charge = ? N2: same logic as N1 if you want to give both oxygens a formal charge of -1 they both need to be single bonds Structure 2: C1: valency of 4; 4 bonds; 0 lone pairs --> formal charge = ?
C1-1 N1-3
N1: valency of 5 - 4 lone pairs = +1 formal charge
on the left structure if you give both oxygens a single bond then the N2 formal charge becomes 0 (3 bonds, 2 lone electrons)
and for c i circled the right one
structure 1: C1 has a -1 formal charge (valency 4, 3 bonds, 2 lone pair electrons) structure 2: C1 has 4 bonds and its valency is 4, so 4 - 4 = 0 formal charge everything else is ok
and i circled structure 2 for c
thanks voca =)
yes that's good
idk if these are the resonances they want but that's my best attempt
Thank you i bet this is right
Join our real-time social learning platform and learn together with your friends!