Which of the following elements in the third period would you expect to have the most negative electron affinity value, and why? Aluminum (Al), because it is a metal with a large atomic radius Chlorine (Cl), because it is a halogen with a high effective nuclear charge Silicon (Si), because metalloids release the most energy when gaining electrons Sodium (Na), because alkali metals only have one electron in the outer energy level
@dude
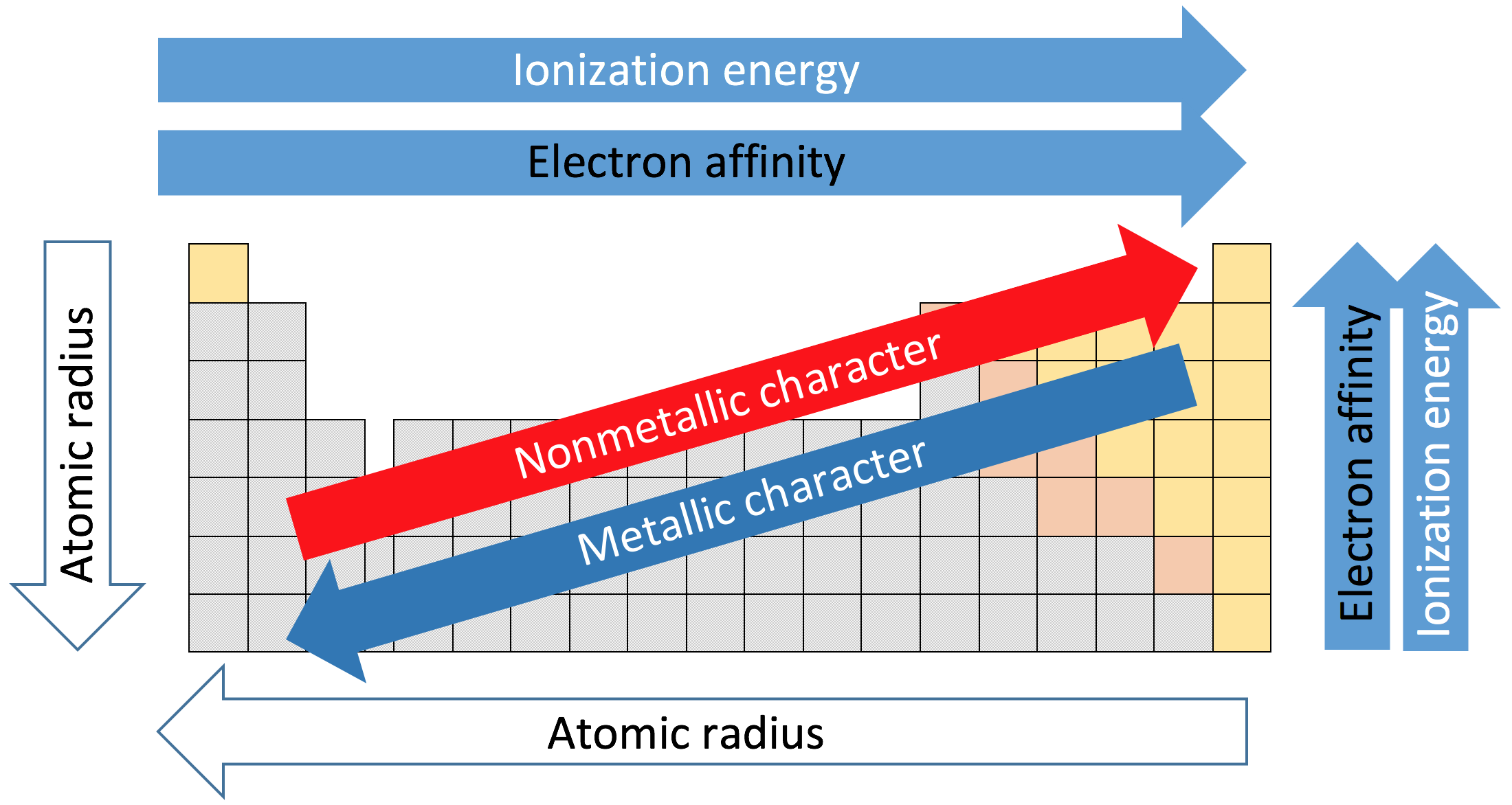
Period 3 is the 3rd horizontal set of elements
So like.... which letter?
I honestly dislike chem so much.
rani lmao you have to try your best and he'll tell u if its right
Okay hold up c?
|dw:1541164464025:dw| Which answer is to the farthest right?
AR?
Yes but its not in the answer choices, which is the next element
AI?
|dw:1541164587312:dw| C'mon
Okay 2 more thoo
OHHHHH
Yes im stupid
Make separate posts
Join our real-time social learning platform and learn together with your friends!
