aldo condensation reaction mechanism
|dw:1561771349638:dw|
Please do not comment unless you are making a serious attempt at a solution.
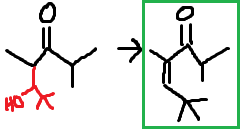
Is this the answer for (c)
Both (c) and (d) are base catalyzed. The mechanism is here https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Supplemental_Modules_(Organic_Chemistry)/Reactions/Organic_Reactions/Aldol_Condensation For (c) we have the aldehyde and it has a tertiary carbon substituent so it doesn't have any hydrogens to react. So you have to convert the other structure to aldol
I put the original aldehyde structure in red just so you could see where it was originally
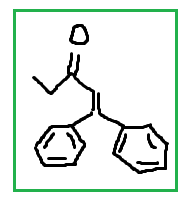
(d)
tbh mechanisms are too detailed for me, I really hope your class doesn't require you to memorize all the mechanisms. Mechanisms are important but there are just too many reactions to know already The way I see aldol-condensation to find the product easily is: the carbonyl of the second structure, just connect where that O is to the alpha carbon of the first structure so you can form the alpha, beta-unsaturated aldehyde/ketone
Thank you c:
My pleasure! Hopefully my answer was of some use :)
Join our real-time social learning platform and learn together with your friends!