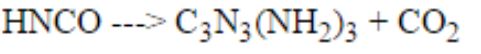What coefficient would go in front of HNCO to balance this equation? the pic is in the comments
\[\rm Left ~side: \]\[\rm H: 1\]\[\rm N: 1\]\[\rm C: 1\]\[\rm O: 1\] \[\rm Right ~side: \]\[\rm H: 6\]\[\rm N: 6\]\[\rm C:4\]\[\rm O: 2\] The number of element on reactant side should be equal to number of element on product side.
btw |dw:1575775721996:dw| we multiply each element inside the parenthesis by 3
So my answer choices are 3,4,5,6 and thank you for the help! But what I dont get is that I can only put on the left side one number to balance it, is that even possible?
a coefficient and plus in order to balance it out you can put a subscript
I know its not 6 because my friend had this before and she got 6 wrong so the answer can be either 3,4,5, or none of the above and its multiple choice so I cant add anything
for this type of questions you have to go back & forth to make sure all the elements satisfied with that one coefficient
lets find out why it is not 6.
In order for it be a 6,I think there has to be a 3 in front of CO2
And I cant do that
|dw:1575776257877:dw| if you put 6 as a coefficient that means all the other elements are also 6 H:6 N:6 C:6 O:6
Yes
Since the question is asking for coefficient for left side that means you can't make a change on the right side.
yeah i cant
so how would you get 2 O on the left side?
I cant the choices dont give me an option, I am wondering now if its a mistake the teacher did.
lets forget about the answer choices for a second. I am trying to help you understand the method so you can do the rest of the questions on your own.
Oh okay well if I were to put a 6 on the left side in order to balance it out I would have to add a 3 in front of CO2|dw:1575776718667:dw|
that's correct. But for this question, we can't change anything on the right side. we need to make all the elements on the left side equal to the number of the elements on the right side. So how would you get H: 6 N: 6 C: 4 O: 2
one more hint: since all the elements are not equal to 6, we need to put some subscript.
Okay so if we did with subscripts wouldnt it be|dw:1575777160259:dw|
|dw:1575777226558:dw|
that's correct!
Thank you for the help! Now I need to talk to my teacher why its like that
like what ?
why the answer choices dont match up
what do you have for the answer choices ?
3,4,5,6 and no coefficient needed
oh okay one second.
okay
yeah confirm with the teacher to make sure there is no typo The wording of the questions is like they are just asking to find a number that will balance the equation without even messing up with the right side. But lets say if we put 3 in front of Co2 and 3 in front of HNCO and then put a subscript of 2 for all the elements \[\rm 3 H_2N_2HC_2O_2\] but doesn't seems right to me
Yeah because it wouldnt work for Oxygen, but thank you for helping me understand this! Much appreciated:)
it will work for oxygen. O will be 6 on both sides.\[\rm 3H_2N_2C_2O_2 \rightarrow C_3N_3(NH_2)_3 +3CO_2\]
\(\color{#0cbb34}{\text{Originally Posted by}}\) @mimih Yeah because it wouldnt work for Oxygen, but thank you for helping me understand this! Much appreciated:) \(\color{#0cbb34}{\text{End of Quote}}\) my pleasure!
Oh okay thanks anyways!
\(\color{#0cbb34}{\text{Originally Posted by}}\) @mimih Oh okay thanks anyways! \(\color{#0cbb34}{\text{End of Quote}}\)
Join our real-time social learning platform and learn together with your friends!