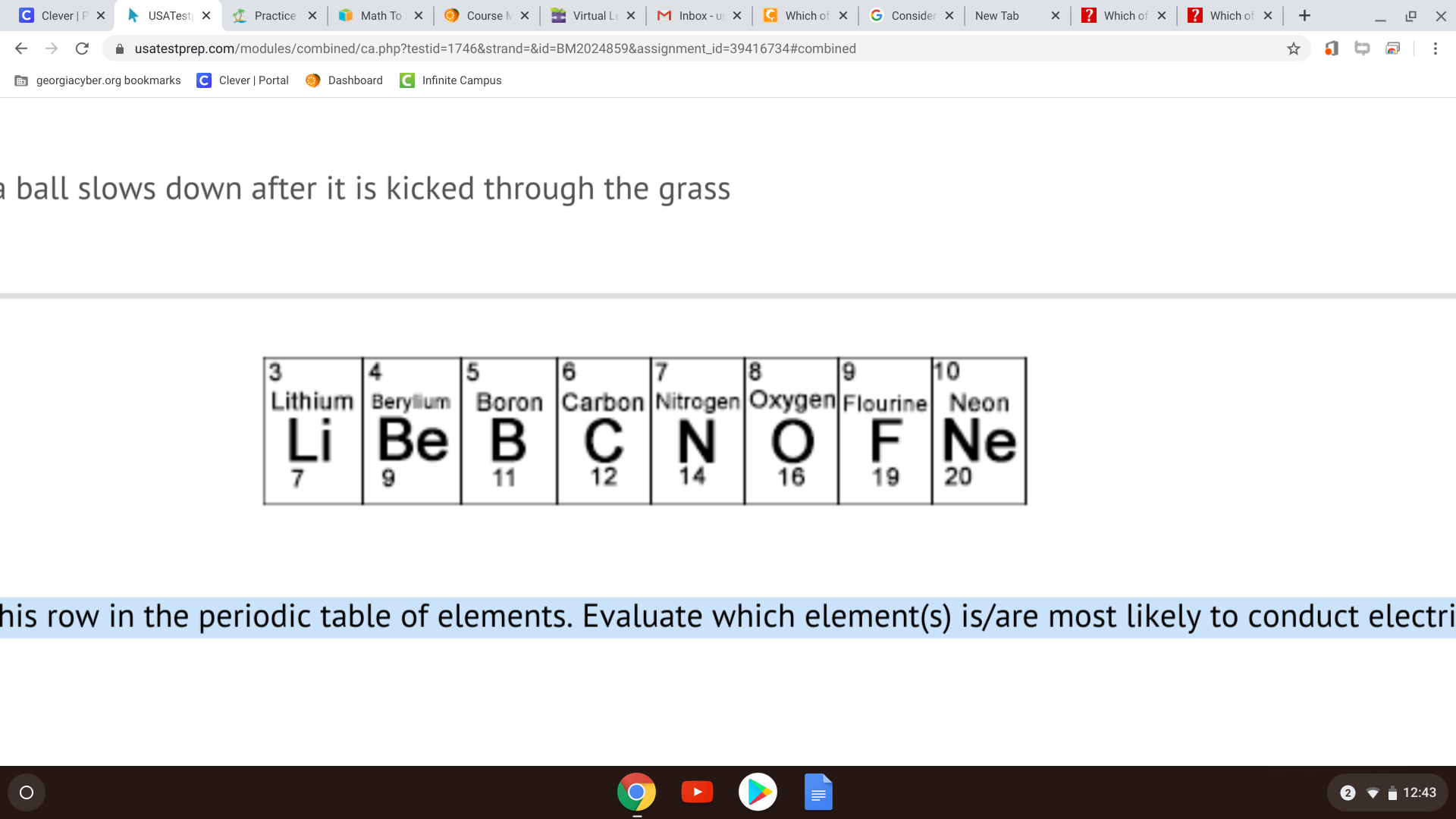
Consider this row in the periodic table of elements. Evaluate which element(s) is/are most likely to conduct electricity. A) lithium and beryllium B) fluorine and neon C) boron and carbon D) lithium
can you help? on this one
........... no clue
Really the hint is that they are both metals
idk lol
lithium?
which options are metals? Look at the periodic table |dw:1582220894653:dw|
Lithium and...
beryllium
yes, correct. it is option A
this one?
And you want to understand why? First, Neon has a valency of 0, and its orbitals are full, so it cannot accept more electrons. Then, it is not boron, carbon, nitrogen, flourine, etc. because even though they are able to form ions (i.e., F-, O2-, etc.), they have a very high ionization energy, so it is difficult for them to keep passing on electrons back and forth. Meanwhile, metals that are on the far left are great conductors because they can ionize and more importantly do not require a strong ionization energy to pass on free electrons
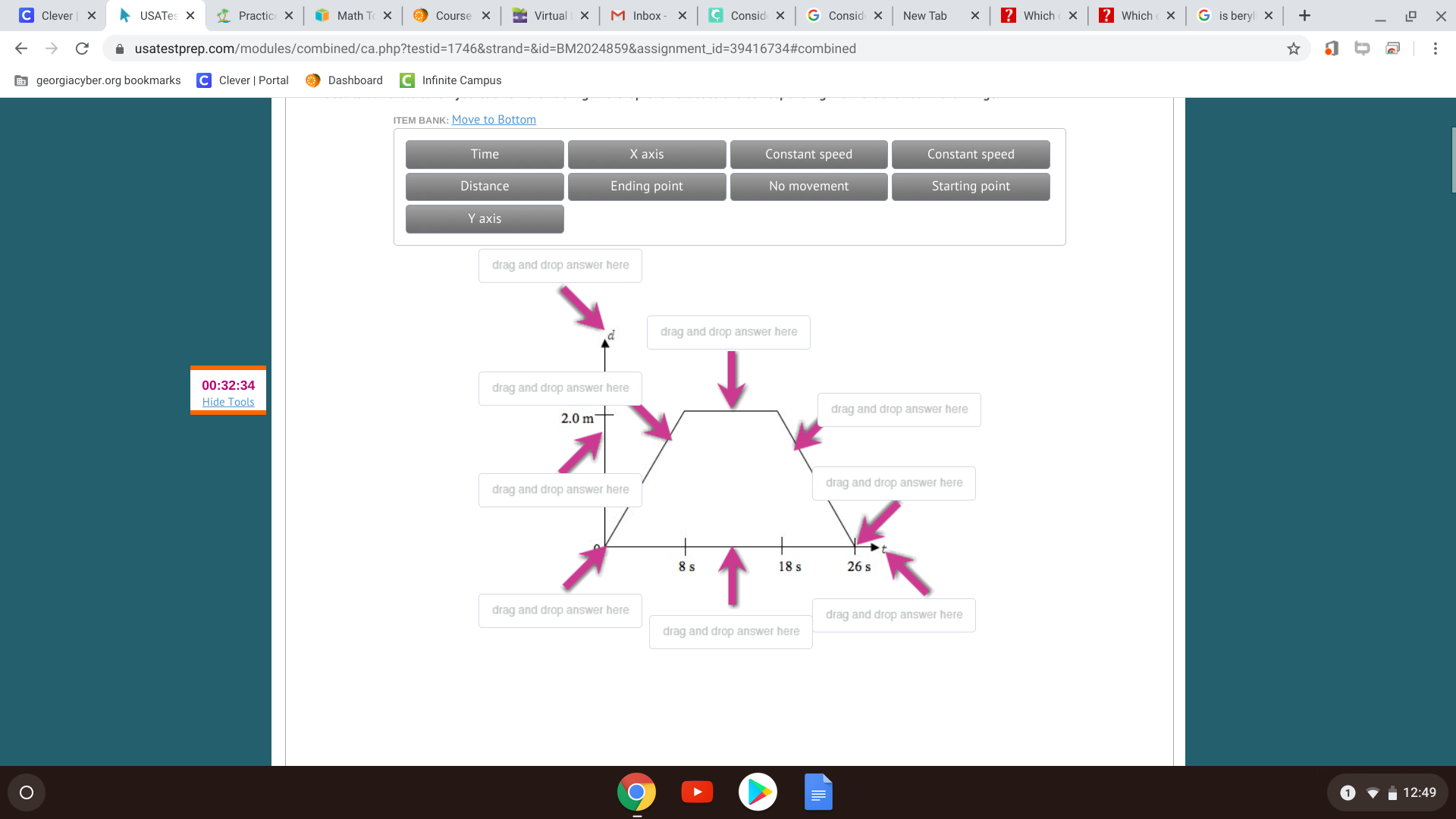
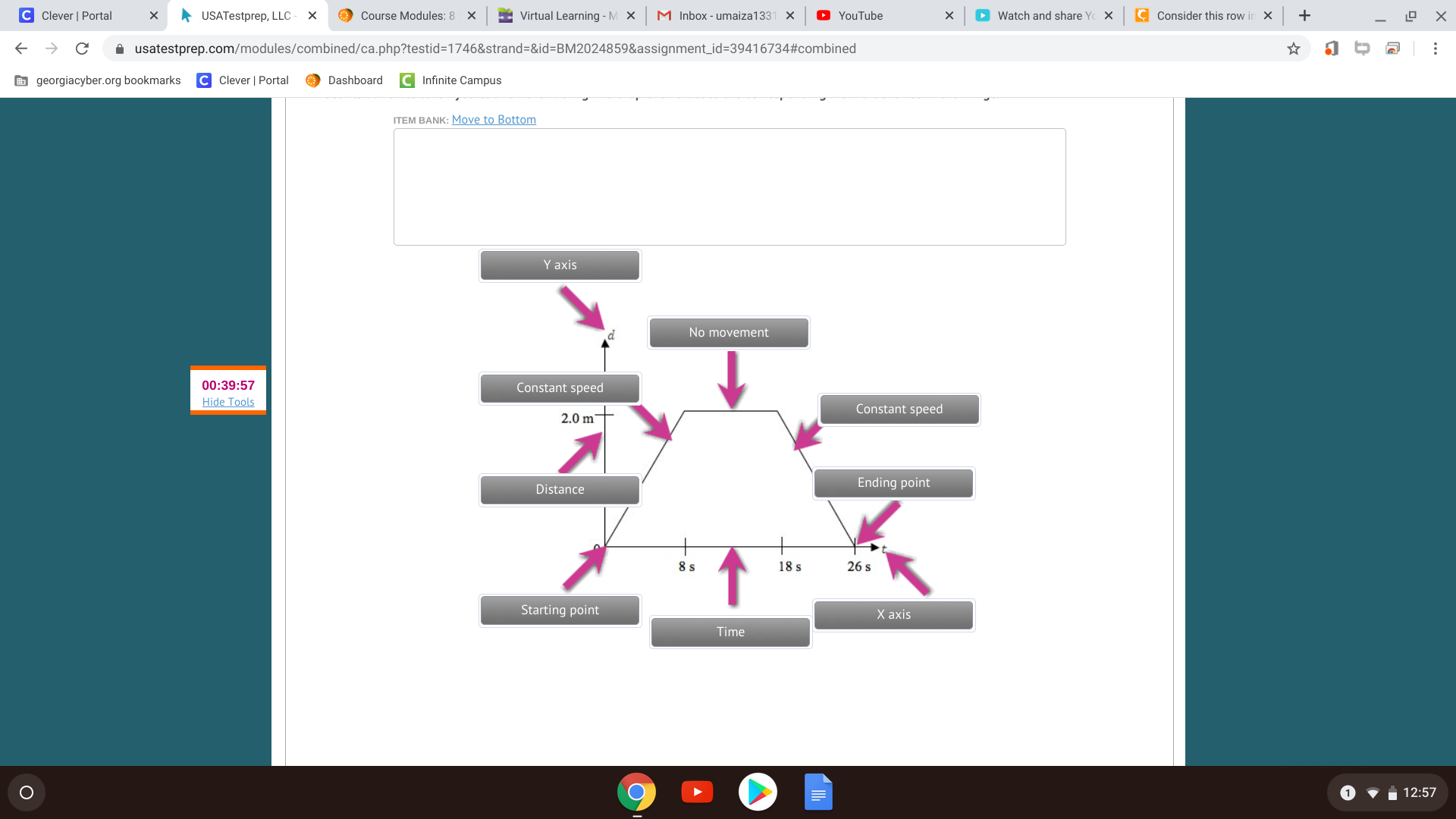
Bruh I think you should be able to do that diagram
How about you fill up what you know and I can check (:
ok
all right but maybe you would want to point distance to the 'd' and time to the 't' and switch the x and y axis alternatively, if that does matter.
nope it doenst
right
ok
yes
ok
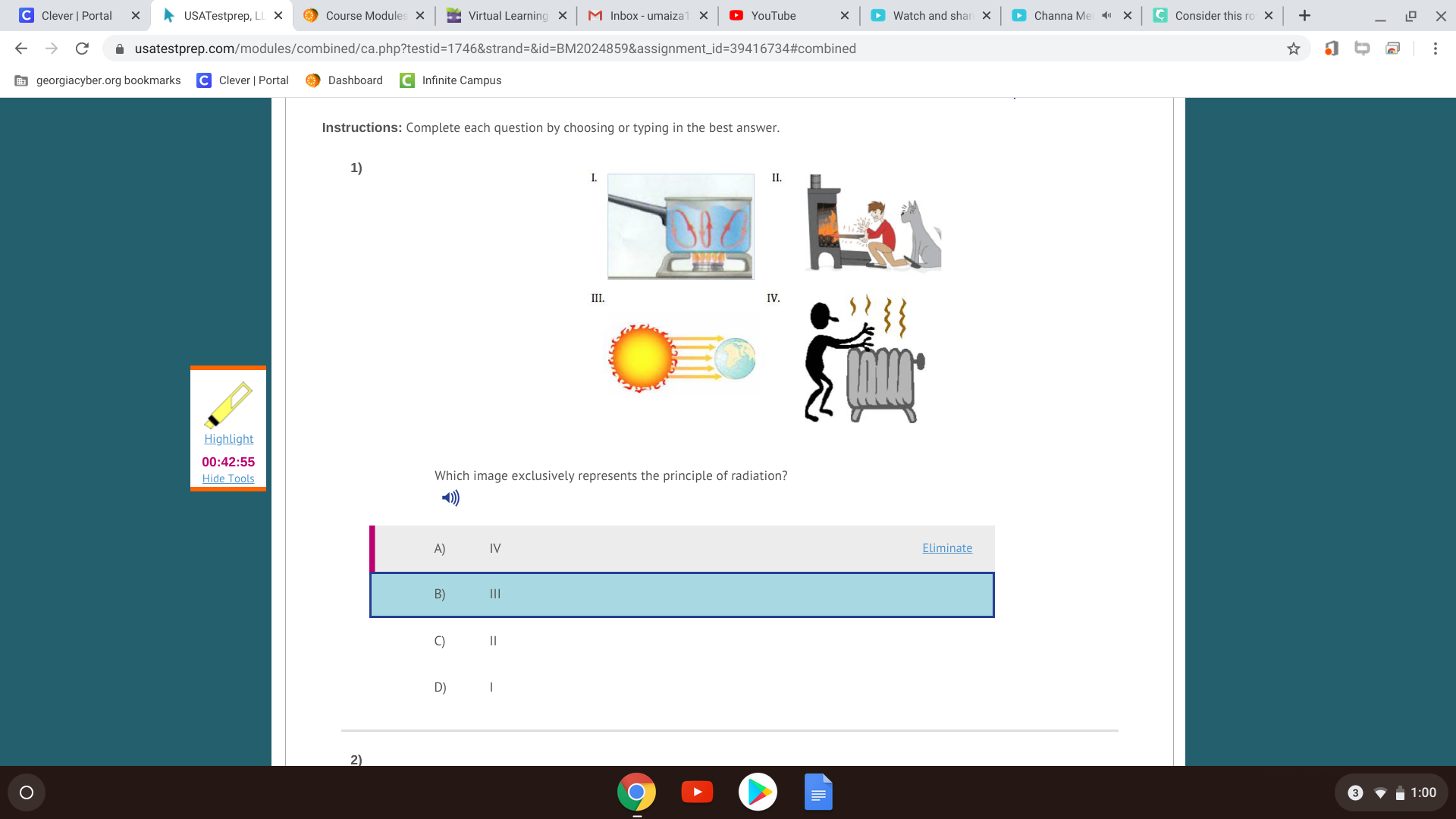
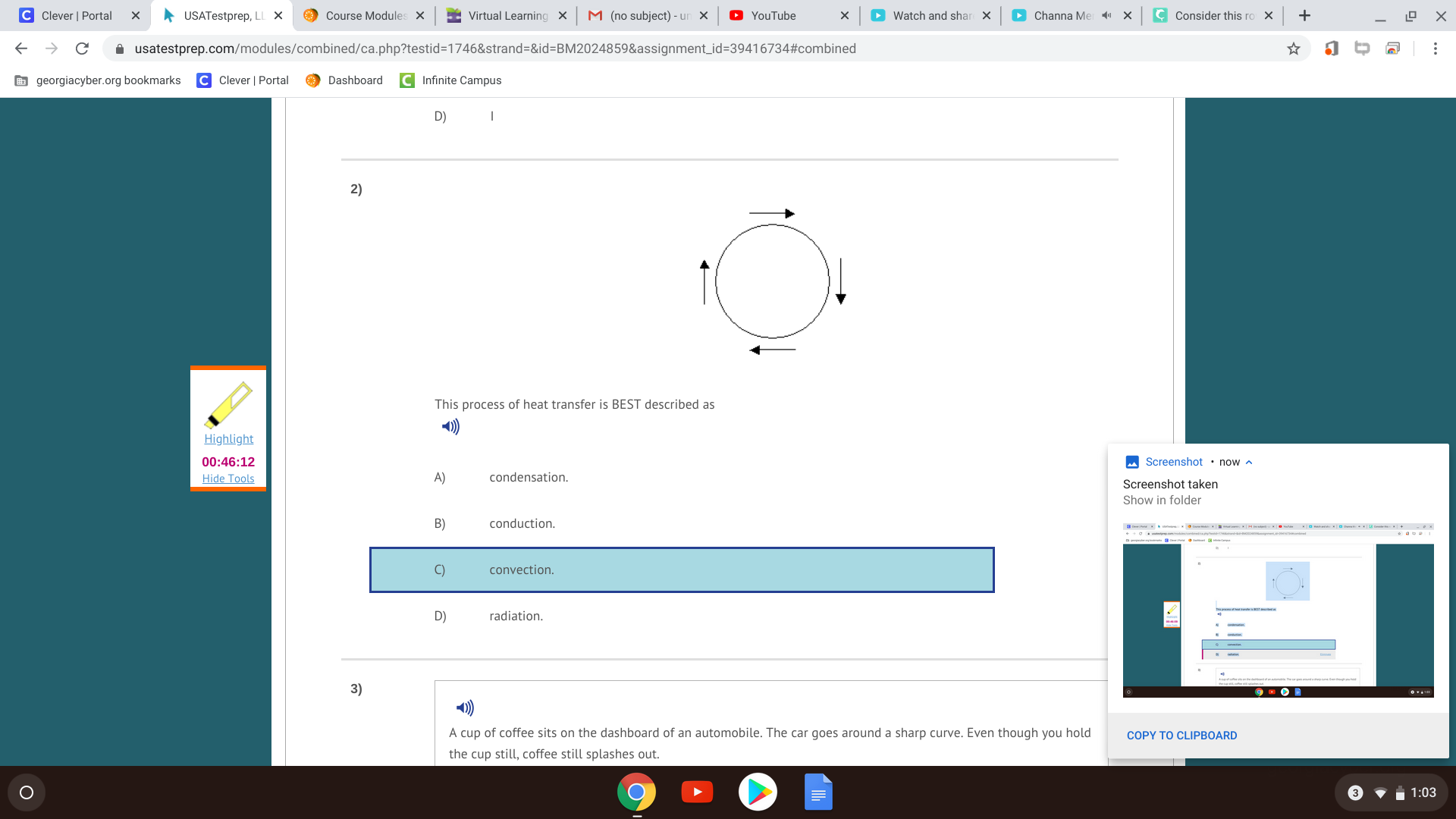
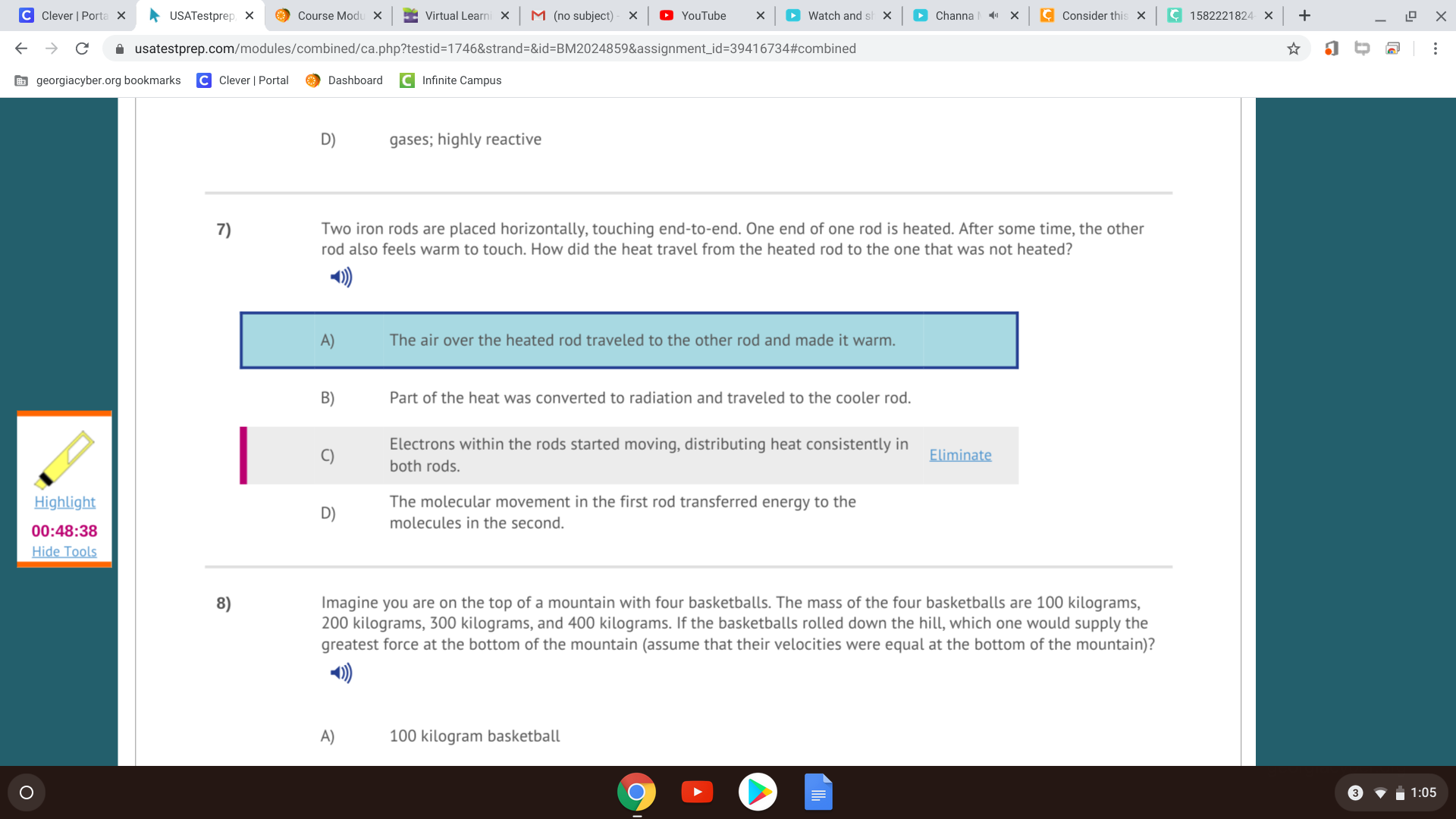
Since they are touching, the heat traveled via conduction. The fast-moving molecules of the warm object collide with the molecules of the cooler one, and they have an elastic collision so that the momentum transfers over, which increases the molecular speed of the molecules in the cooler object.
so its wrong wich one is right then?
The molecules in one object transfer energy to the other
dude I can't keep answering your questions, maybe someone else can help you
D?
yes
maybe post some of your questions as a new post so more people can see it and help
Join our real-time social learning platform and learn together with your friends!