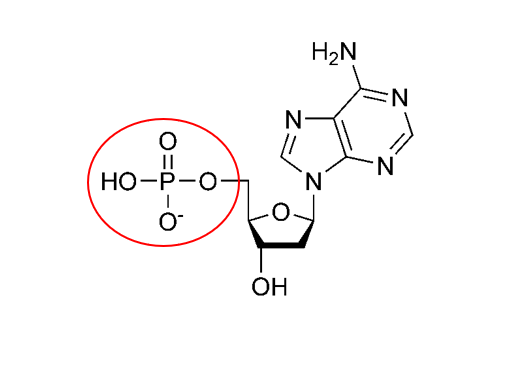
what functional group is this
im thinking phosphate but im not sure
@Vocaloid
yup, that's a phosphate group, good work https://www.thoughtco.com/thmb/YTAtfLqCD0slvOgPAhXC14oFKXk=/1500x1000/filters:fill(auto,1)/what-are-the-parts-of-nucleotide-606385-FINAL-5b76fa94c9e77c0025543061.png
i have one question regarding it i read that it needs two neg oxygen if its positioned at the end and one if its inside a chain of phosphates and that applies here correct?
hmm I wouldn't necessarily agree with that, whether a phosphate group has protons (H atoms) on the oxygens depends more on the pH of the enviornment I'll do a little reading to confirm
ah, ok, they're referring to how oxygen acts as an electron donor within a chain of phosphates, and thus the overall molecule "loses" a negative charge |dw:1599147065845:dw|
just in case the image doesn't show up https://lh3.googleusercontent.com/proxy/jFsnD1rdv75tZbLmqLpXl0FSqmkEUs4Bc1t2ufnf6rUmgs5pJ6DZ4vdZOrdBtlVMwrN_8uToMh9LmbpH_cPY2fN0rROmeoj4E-_VeQ
oh i see , thank you very much!
Join our real-time social learning platform and learn together with your friends!