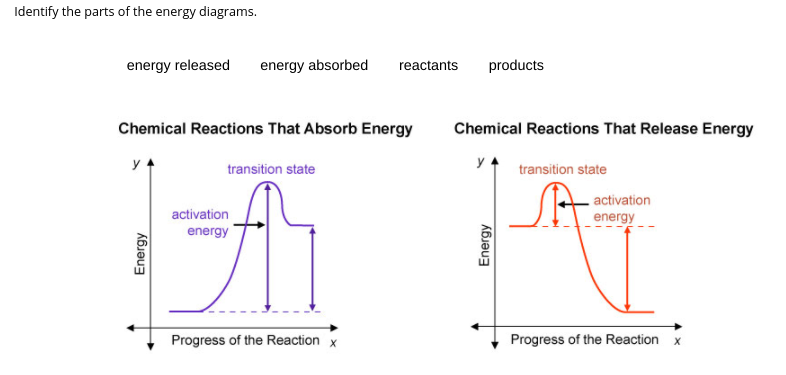
The first comment is the question I need help answering.
Here it is
Please help me if you figure out the answer. :)
You're going to need to know that reactions go from reactants
> products so your graphs are going to begin with your reactants and they'll finally end up as your product
Oh okay, thank you.
oops I don't know why it did that but it should be reactants ---> products
as for energy released and energy absorbed if your reactants have MORE energy than your product, you would have RELEASED energy if your reactants have LESS energy than your products, you would have ABSORBED energy.
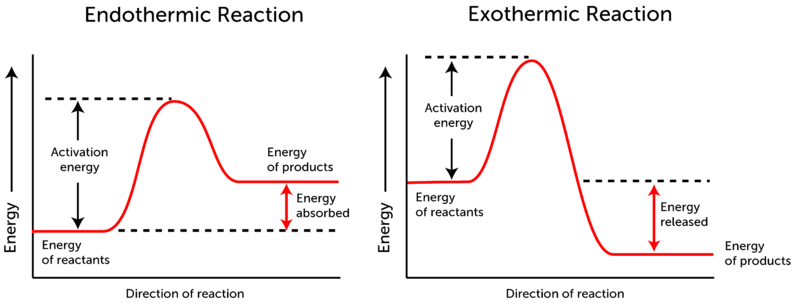
Here's a nice graph that shows you it all. You should get very familiar with this image and the concepts it represents
Okay Thank you :)
No problem! Let me know if you have any questions or if you want me to check how you've labeled it!
ok!
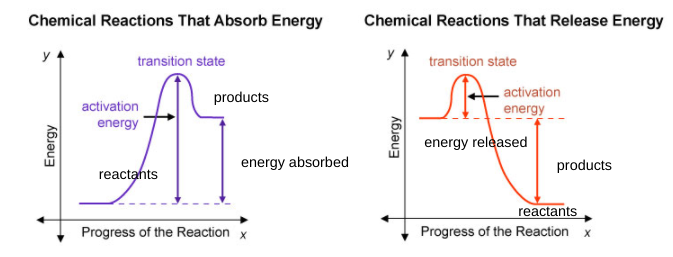
@AZ is this correct?
well the reactants on the picture on the right is where the products should be but other than that, it looks good
so I should just swap them?
uhh swap? You put but the reactants and products on the right side Take a look at the image I attached earlier. Remember that the graph starts off as reactants and ends as product
OH! I get it thank you for helping me!
Have a great day, bye!
No problem! You too!
Join our real-time social learning platform and learn together with your friends!