Can I get some chemistry help?
What kind of chemistry?
An exothermic reaction releases heat so the equation would be \(\sf SO_2 (g) + Cl_2(g) \to SO_2Cl_2 (g) + heat\) Do you know what Le Chatlier's principle is?
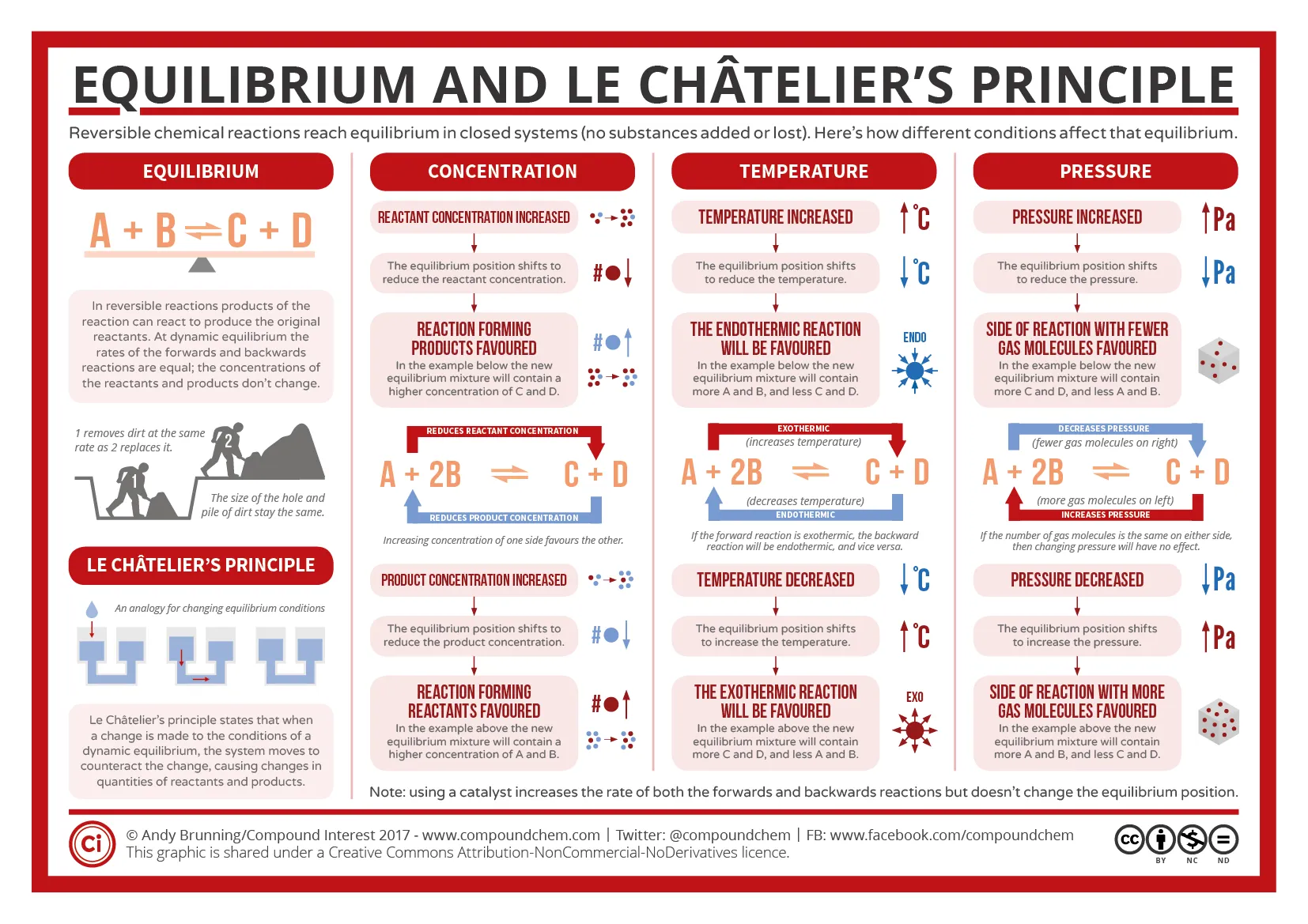
There's also this very nice image So we want to shift the reaction to the left. Which of your options would do that?
Increasing the pressure?
I know its pressure or temperature but I am a bit confused
Good, you've eliminated it down to one of the correct answers :) When you increase the pressure with gases, it's going to shift towards the side with FEWER moles you can see on the left side that you have 2 moles- SO2 and Cl2 On the right side, you have 1 mole- SO2Cl2 so which way would the reaction shift?
Right with increased pressure, left with temperature so temp would be the answer
Exactly!
Could you check some of my answered questions for me?
Sure, tag me in your next post :)
Join our real-time social learning platform and learn together with your friends!