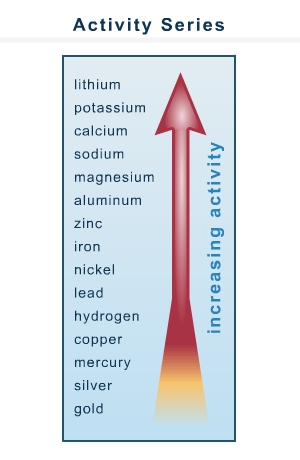
Which of these redox reactions is spontaneous as written? Ni + Zn2+ → Ni2+ + Zn Mg + Li+ → Mg2+ + Li Cu + Au3+ → Cu2+ + Au Zn + Ca2+ → Zn2+ + Ca
It's for my final.
I am sorry to say but I have no Peanut Buttering idea. Im 14 so......
that's fine. Thank you anyway.
hope you do good on your final.
Thanks. Only 300 more questions total before Friday....
Oh hell no
That's how I feel..
Well I hope you survive.
Thank you lol
can you take the screenshot of the question ?
I can try.
Which of these redox reactions is spontaneous as written? Ni + Zn2+ → Ni2+ + Zn Mg + Li+ → Mg2+ + Li Cu + Au3+ → Cu2+ + Au Zn + Ca2+ → Zn2+ + Ca
That's the best I could do.
yeah that picture is important!!! without you can't find the answer
it depends on which element is more active the more active element will ions in the solution(product)
I found it already. I kinda guessed lol
which one ?
I don't remember, but I got it right.
Join our real-time social learning platform and learn together with your friends!