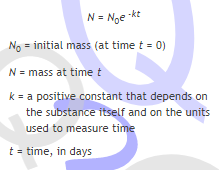A 15-gram sample of a substance that's a by-product of fireworks has a k-value of 0.1405. Find the substance's half-life, in days. Round to the nearest tenth.
"half life" is the amount of time it takes for a substance to lose 50% of it's mass so we use our formula y = a(e^kt) and plug in 7.5 for y, 15 for a, and 0.1405 for k
7.5 = 15 * e^(0.1405)t dividing both sides by 7.5 gives us 0.5 = e^(0.1405t) know where to go from here?
no idea lol i suck at this
in order to "eliminate" the e^( part we must take the natural log of both sides ln(0.5) = lne^(0.1405t) if we have ln(e^something) we can cross out the ln(e part so we get ln(0.5) = 0.1405t then we simply divide both sides by 0.1405
I get 4.9 days
oh... im still so lost lol
that was right tho :D
well, let's take a closer look at our formula
y = a(e^-kt) *I forgot to put the negative sign before
this is the formula they give
ooh ok
since your problem gives you: the initial mass the final mass and k, you can insert these into the formula and find t
|dw:1501390253430:dw|
wait whats the final mass?
|dw:1501390271584:dw|
since they are asking for "half-life" the final mass = initial mass/2
half-life is the amount of time it takes for the final mass to equal half the intial mass
i feel really dumb rn lol
don't worry about it, it's just a definition to memorize
wait than whats the 0 for?
the "0" is a subscript, it's just to make the N_0 different from the other N
oh ok
it's just a fancy label, it doesn't get treated like an actual 0
any other questions?
prob
i have another one can u like guide me through it?
sure, you can close this question and make a new one
cool
Join our real-time social learning platform and learn together with your friends!