Ask
your own question, for FREE!
Chemistry
21 Online
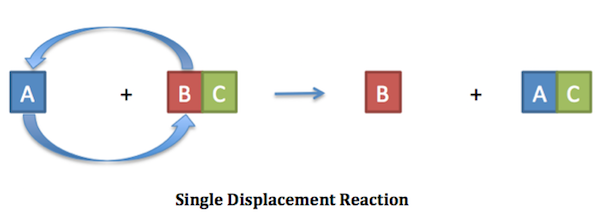
Study this equation carefully. What classification should this reaction have? Cu + 2AgNO3 Cu(NO3)2 + 2Ag
Still Need Help?
Join the QuestionCove community and study together with friends!
This is a double displacement reaction. This reaction occurs because copper is more reactive than the silver which is in solution as silver nitrate
Should be single replacement actually Copper would need to be in an ionic compound to be double replacement
Moved to the Chemistry section from Mathematics.
Closed due to answered.
Can't find your answer?
Make a FREE account and ask your own questions, OR help others and earn volunteer hours!
Join our real-time social learning platform and learn together with your friends!
Join our real-time social learning platform and learn together with your friends!
Latest Questions
 Twaylor:
how to make good breakfast food ingredients : 1 mother flat bread bananas peanut
Twaylor:
how to make good breakfast food ingredients : 1 mother flat bread bananas peanut
 lovelove1700:
u00bfA quu00e9 hora es tu clase?Fill in the blanks Activity unlimited attempts left Completa.
lovelove1700:
u00bfA quu00e9 hora es tu clase?Fill in the blanks Activity unlimited attempts left Completa.
 glomore600:
find someone says that that one person your talking to doesn't really like you should I take their advice and leave or should I ask the person i'm talking t
glomore600:
find someone says that that one person your talking to doesn't really like you should I take their advice and leave or should I ask the person i'm talking t
 Addif9911:
Him I dimmed the light that once felt mine, a glow I never meant to lose. I over-read the shadows, let voices crowd the room where only two hearts shouldu20
Addif9911:
Him I dimmed the light that once felt mine, a glow I never meant to lose. I over-read the shadows, let voices crowd the room where only two hearts shouldu20
 EdwinJsHispanic:
Poem to my mom who proved my point "You proved my point, I am a failure. but I kinda wish, you were my savior.
EdwinJsHispanic:
Poem to my mom who proved my point "You proved my point, I am a failure. but I kinda wish, you were my savior.
 Wolfwoods:
The Modern Princess "you spoke so softly to me, held me close when no one else did, loved me in a way no one else dared to.
Wolfwoods:
The Modern Princess "you spoke so softly to me, held me close when no one else did, loved me in a way no one else dared to.
 Wolfwoods:
The Pain Of Waiting "The short story would be that we fell in love, you left and I continued to wait for you.
Wolfwoods:
The Pain Of Waiting "The short story would be that we fell in love, you left and I continued to wait for you.
1 hour ago
0 Replies
0 Medals
43 minutes ago
2 Replies
0 Medals
22 hours ago
5 Replies
0 Medals
2 days ago
4 Replies
0 Medals
2 days ago
6 Replies
2 Medals
3 days ago
11 Replies
3 Medals
3 days ago
7 Replies
2 Medals