help please
@Vocaloid
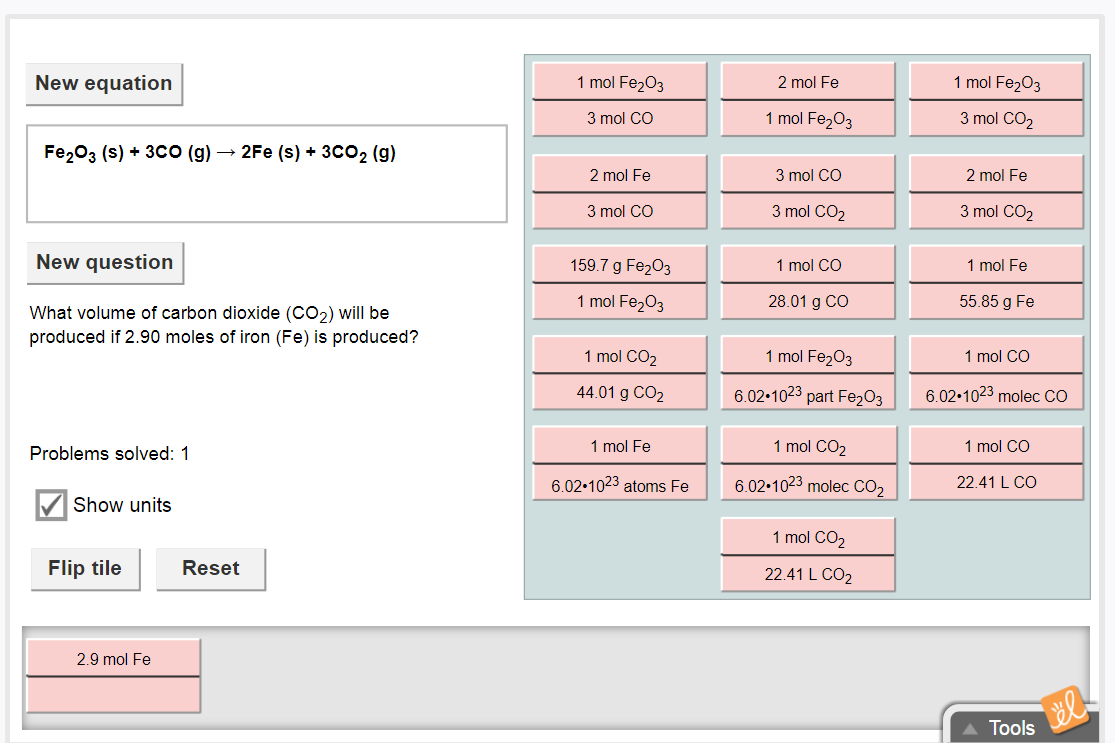
since we want to convert from moles Fe to moles CO2 pick the tile that has the ratio between these two compounds, in moles only
it's the tile that has moles Fe and moles CO2.
so something with 3?
|dw:1517441663639:dw|
|dw:1517441668606:dw|
since you have 2.9 mol Fe in the numerator, you need to also flip the Co2/Fe tile so that moles of Fe are in the denominator, which will allow us to cancel out the units of Fe|dw:1517441715152:dw|
|dw:1517441736657:dw|
since we are now in moles CO2 and we want volume of CO2, select the tile that gives you the ratio between moles CO2 and volume CO2
select the tile that has moles of CO2 and volume of Co2.
okay
i did and it crossed it out
that's the tile that needs to be added to the answer at the bottom you should have gotten 1 mol CO2/22.4L CO2 as the tile now we flip that tile to get 22.4L CO2/1 mol CO2 so we can eliminate the units mol CO2 and get L CO2
|dw:1517442241615:dw|
the tiles are now in the correct order, to get the numerical answer just multiply the numerators and then divide by the denominators so 2.9 * 3 * 22.4/2 ---> simplify to get the numerical answer
i got 97.44
that right?
good, and if they care about significant figures round that to 97 L (since we are only given 2 sig figs for 2.9)
they just want the answer unit and the solution
go with 97 L then
next question?
sure
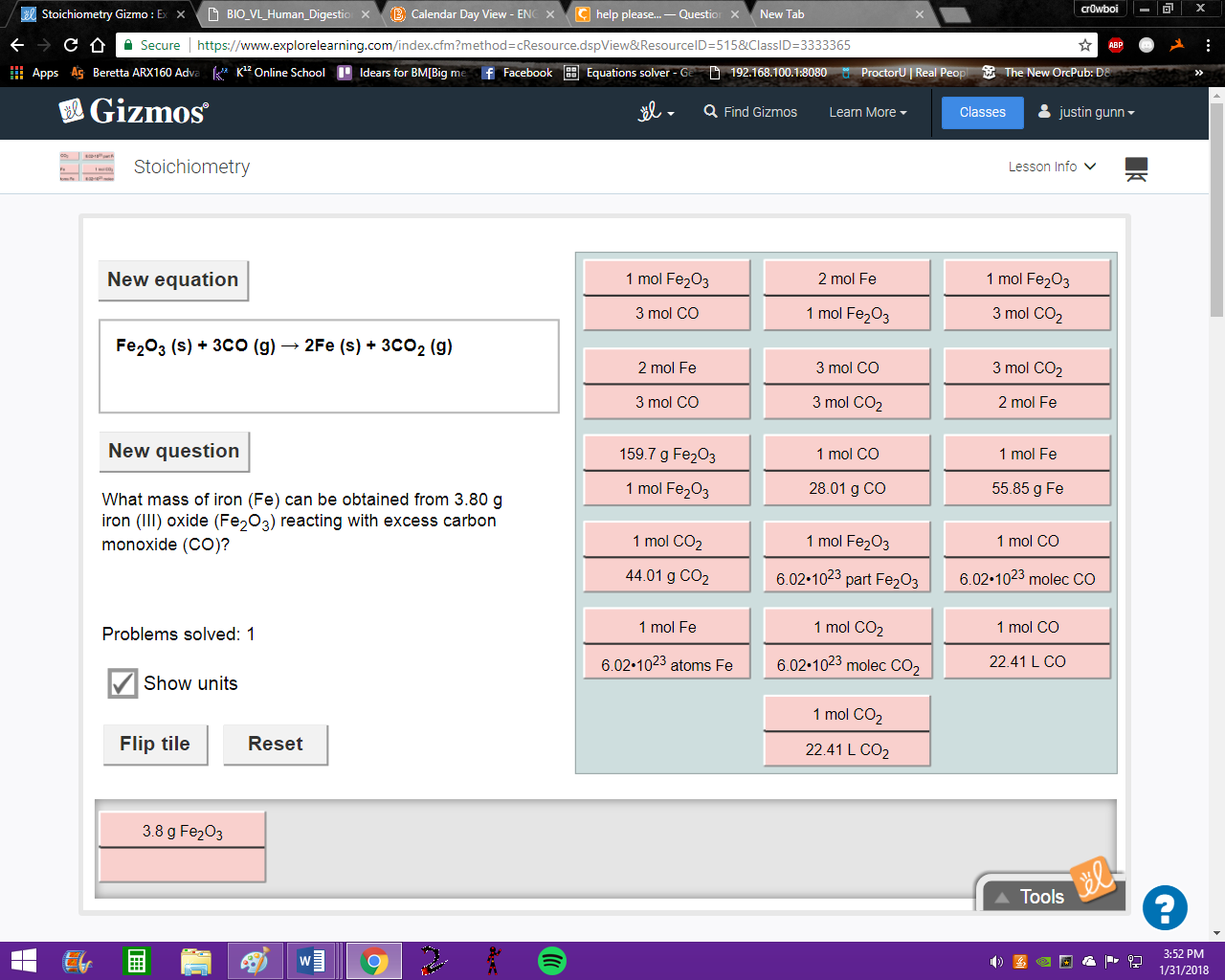
first step is to convert from g iron (III) oxide to moles iron (III) oxide to select the appropriate tile for these two quantities
*so select
that tile would be?
didnt know why that took so long to load..
\(\color{#0cbb34}{\text{Originally Posted by}}\) @Vocaloid first step is to convert from g iron (III) oxide to moles iron (III) oxide to select the appropriate tile for these two quantities \(\color{#0cbb34}{\text{End of Quote}}\) the tile needs to have both moles of iron (III) oxide and g iron (III) oxide, since we are converting between these two values
the two quantities we want are moles iron(III) oxide and grams iron(III) oxide select the tile that contains these two quantities.
the top middle one?
those are both moles one side needs to be moles and the other side needs to be grams
ah, third row third down?
both of them need to be iron (III) oxide not just iron as a reminder the formula for iron(III) oxide is Fe2O3
first row third down?
thats gotta be it
awesome, now since we have 3.8g iron (III) we need to flip that tile so that grams of iron (III) is in the denominator and we can cancel out units
|dw:1517444070311:dw|
|dw:1517444076985:dw|
now we need to go from moles Fe2CO3 to moles iron (Fe) so we need the tile with moles Fe + moles Fe2CO3
that was the top middle one right?
good, since the moles of Fe2CO3 are in the denominator in the tile we don't have to flip this one
|dw:1517444268245:dw|
now we just need to go from moles Fe to g Fe, select the right tile
a fourth one?
third row third across
|dw:1517444365141:dw| we need to flip it so units cancel
so we just multiply/divide as if these were just normal fractions so (3.8 * 2 * 55.8)/159.78 ---> simplify and that's it
okay, i got it, 2 more questions
sure
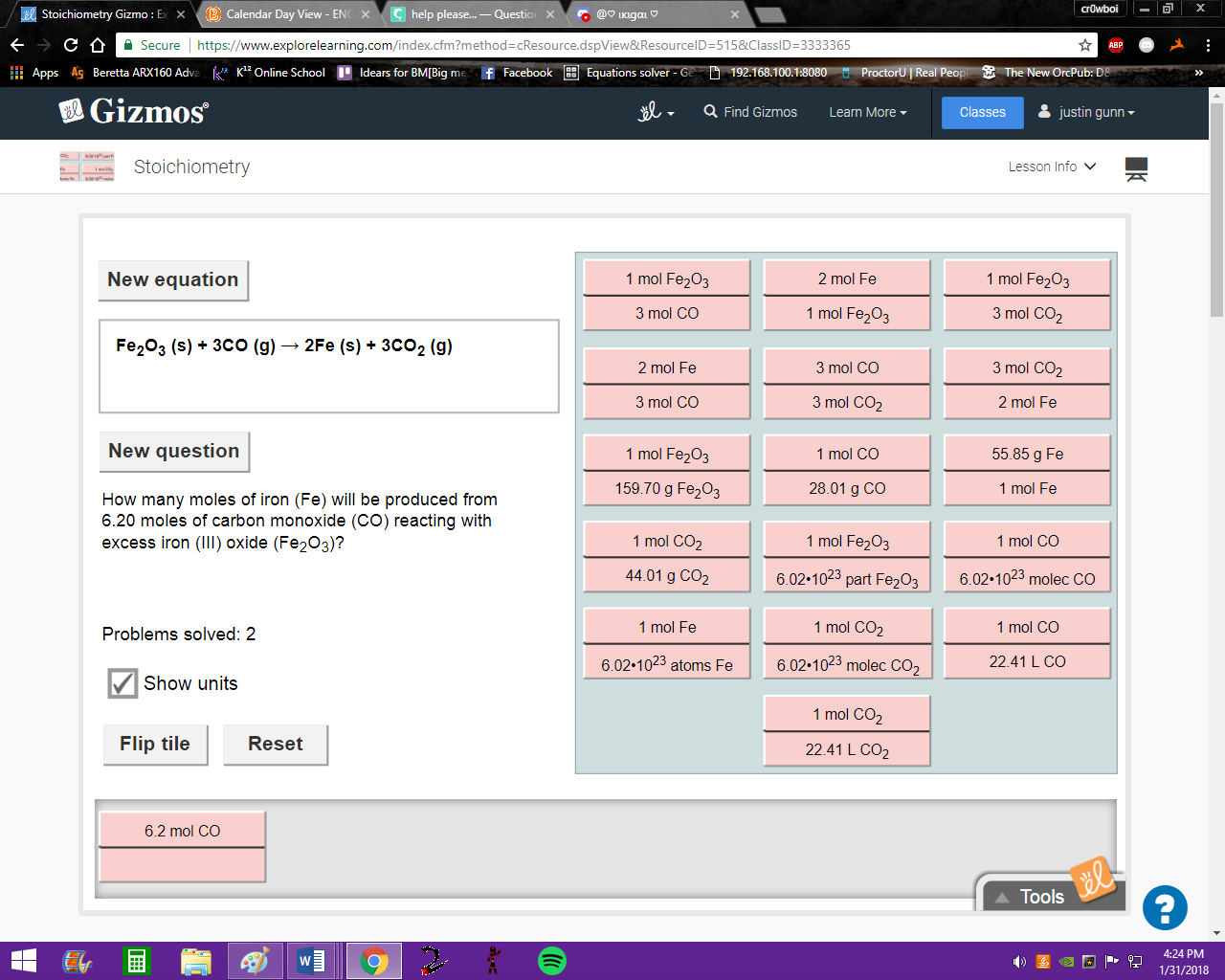
this is a simpler one moles CO --> moles Fe so select the tile that has both these quantities
first row second down
oh, that was it.
okay, last question
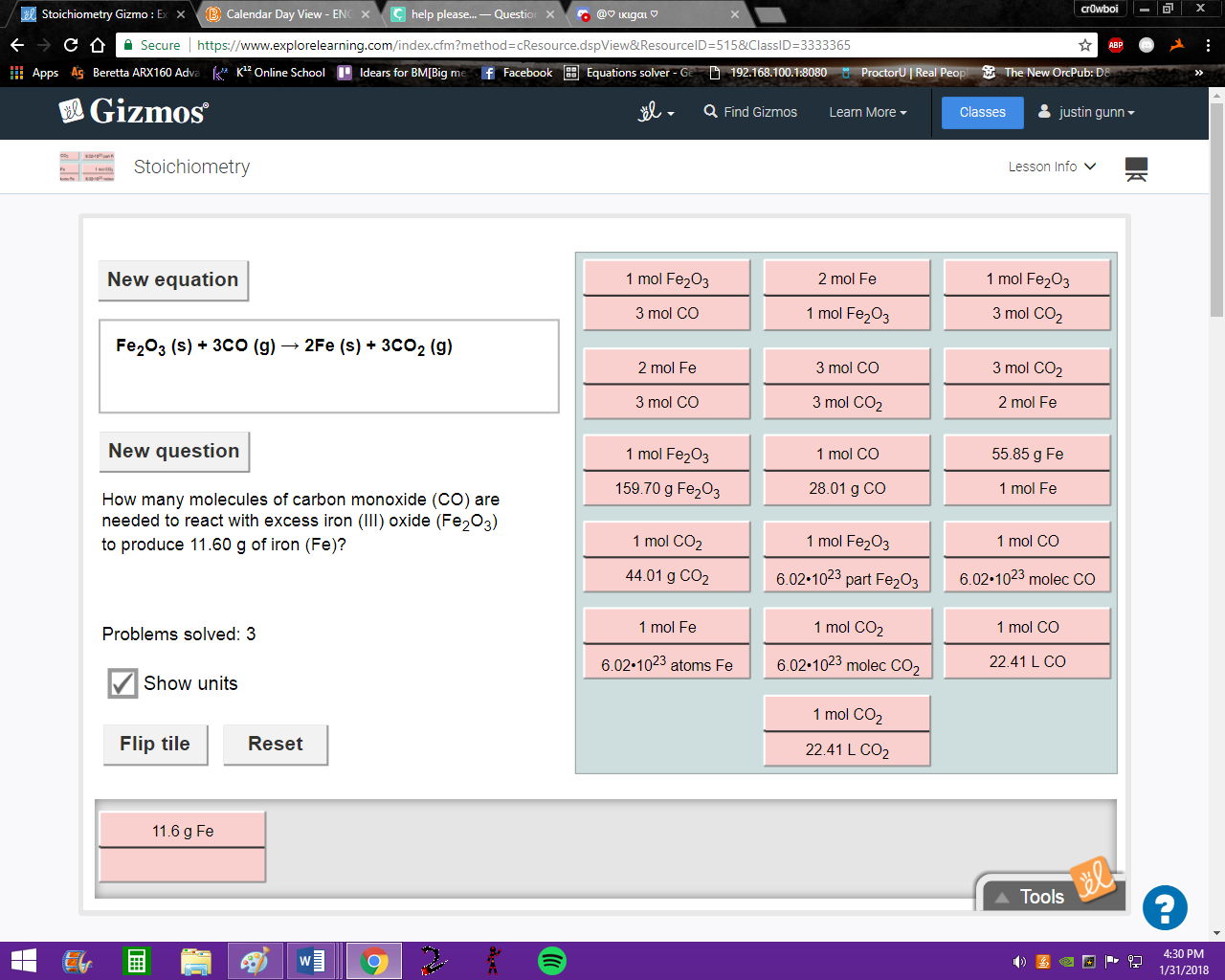
first step is grams Iron (Fe) to moles iron (Fe) ---> pick the tile
third row third down
good, and we need to flip that tile so grams are in the denominator
|dw:1517445333086:dw|
|dw:1517445338411:dw|
now, moles Fe --> moles CO select the appropriate tile
first row second down
good, now we flip that to get |dw:1517445457998:dw|
now it's moles CO --> molecules CO --> tile?
okay
if you want to help me with more, that would be nice, but it's okay if 4 was enough
I'm still free, if you still need help I'm willing to help
okay, i might need to make a new question
Join our real-time social learning platform and learn together with your friends!