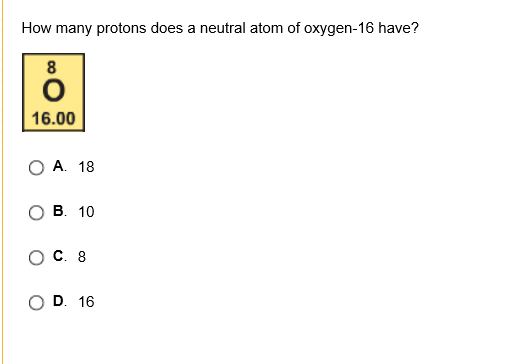Physics
18 Online
zarkam21:
Help please
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
8 years ago
zarkam21:
@Vocaloid
8 years ago
Warriorz13:
I want to go with C but not 100% sure
8 years ago
zarkam21:
well we can double check with @Vocaloid
8 years ago
Vocaloid:
according to the plum pudding model all the alpha particles should have passed through the electron b/c the electron charge is evenly distributed, however some were deflected proving that negative charges were not distributed evenly
which answer choice best reflects this idea?
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
A
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
Vocaloid:
remember rutherford's gold foil experiments - if the alpha particles deflected off the center what does that say about the atom?
8 years ago
zarkam21:
C
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
D?
8 years ago
Vocaloid:
neutrons are neutral
8 years ago
zarkam21:
oh i got confused i thought electrons were
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
so A
8 years ago
Vocaloid:
yes
8 years ago
zarkam21:
8 years ago
zarkam21:
C
8 years ago
Vocaloid:
good
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
8 years ago
zarkam21:
D
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
B
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
C?
8 years ago
zarkam21:
NO worries, no disturbance ^~^
8 years ago
Vocaloid:
hint - how many protons does chlorine normally have? based on its atomic number
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
17
8 years ago
Vocaloid:
good, and when it loses 1 proton how many protons does it have now?
8 years ago
zarkam21:
116
8 years ago
zarkam21:
16
8 years ago
zarkam21:
SO B
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
Vocaloid:
good, so B not c
8 years ago
Vocaloid:
yes
8 years ago
zarkam21:
8 years ago
zarkam21:
D
8 years ago
Vocaloid:
good
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
8 years ago
zarkam21:
B
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
B
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
Vocaloid:
any ideas? process of elimination can help on this one (i.e d is out because ALL molecules are made of atoms, that doesn't explain anything about forces)
8 years ago
zarkam21:
i think either a or c
8 years ago
Vocaloid:
note - intermolecular forces come from unequal charge distribution (A)
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
8 years ago
zarkam21:
A
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
C
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
Vocaloid:
nope, hint - check the noble gases
8 years ago
zarkam21:
B
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
A
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
A
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
C
8 years ago
Vocaloid:
not quite - check to see which element is the most towards the left side of the ptable
8 years ago
zarkam21:
B
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
A
8 years ago
Vocaloid:
good
8 years ago
zarkam21:
8 years ago
zarkam21:
A
8 years ago
Vocaloid:
not quite, a metallic bond would allow for the transfer of electrons
any other guesses/ideas?
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
eithr c or d :/
8 years ago
Vocaloid:
metals are conductors, not insulators
so c is the best answer
8 years ago
zarkam21:
8 years ago
zarkam21:
D
8 years ago
Vocaloid:
good
8 years ago
Join the QuestionCove community and study together with friends!
Sign Up
zarkam21:
8 years ago
zarkam21:
A
8 years ago
Vocaloid:
nope, H and O are nonmetals so this would be covalent
which compound has a metal + nonmetal bonded?
8 years ago
zarkam21:
B
8 years ago
Vocaloid:
good
8 years ago