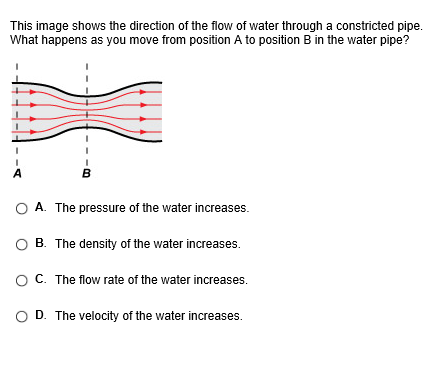
Help please
A?
@Vocaloid
check your calculations again (5/2)nRT = (5/2)(3)(8.31)(25+273) = ?
(5/2)(3)(8.31)(25+273) = ? you just need to chuck this into a calculator ^^
C
good!
C
well done
D
this is a very common mistake but temperature is the measure of avg. molecular velocity, not heat; heat is a measure of thermal energy
so temperature
pressure = Force/Area since we're talking about the same block, force of gravity is equal, so we are looking for the smallest surface area of the side
c
9 * 3 = 27 but there's a smaller one than that
B
good, B
you're given A1, F1, and A2, just need to plug in and solve for F2 F1/A1 = F2/A2 2500/0.06 = F2/0.18 solve for F2
a
good
d
good
B
good
B
good
either a or b
well we determined earlier that Archimedes' principle states that buoyant force is proportional to volume (so A not B)
D
yeah good
B
water molecules are very attracted to each other causing them to bond together on surfaces, resulting in high surface tension not low as a hint, water has a high heat capacity which would result in which answer choice as the correct conclusion?
A
recall that a high heat capacity means that water must take in a lot of heat to change its temperature, the opposite of what A is stating
uugh um d
good, D
Ugh stuck between b and d
B would be cohesion (being attracted to itself), D would be adhesion (attraction to other surfaces), so D
B
good
B
good
well let's start with in nucleus vs. out, there are two particles that are in and one type that is out, what are they?
Actually I did it, can you just check it over
good but you missed "smallest" which particle is this?
oh wait you put it under electron never mind yeah that's right
No i didnt
smallest would be electron right?
yeah good
Compare the numbers of protons and electrons in a positive ion with the numbers of protons and electrons in a negative ion.
well if it's positive which is greater, # protons or # electrons?
protons
good, so positive ion would be "more protons than electrons" and then for negative ion it would just be the opposite, "more electrons than protons" that's really it
How many protons, neutrons, and electrons does a neutral atom of nitrogen-15 have
well, # of protons is easy --> just look at the ptable and find its atomic number
ptable.com atomic number is 7, then the number of electrons is also equal to protons since the atom is neutral, then to find neutrons just subtract mass # (15) - atomic number (7)
A nitrogen atom has 7 protons, typically 7 neutrons, and 7 electron
you're on the right track but we are talking about nitrogen-15 which is not a typical nitrogen atom
# of neutrons = mass # - atomic # = 15 - 7 = ?
8
good so 7 protons ; 7 electrons ; 8 neutrons
Many scientists have contributed to the atomic theory. Match the scientists below with the correct descriptions of their work. Some scientists will be used more than once. (1 point) A. Thomson B. Bohr C. Rutherford D. Dalton _____ Believed that electrons move in specific orbits _____ Discovered electrons _____ Believed that atoms could not be broken into smaller parts _____ Experimented with cathode rays _____ Inferred that atoms have a small, dense, positive nucleus _____ Developed plum-pudding model _____ Shot particles at gold foil
Well the last two are c and a
yeah, that's good, for this one you really just have to look them up ex. electrons in orbits would be Bohr
lmk if you're getting stuck or need help
__B___ Believed that electrons move in specific orbits __A___ Discovered electrons __D___ Believed that atoms could not be broken into smaller parts ___A__ Experimented with cathode rays _____ Inferred that atoms have a small, dense, positive nucleus ___C__ Developed plum-pudding model __A___ Shot particles at gold foil
you mixed up the last two, rutherford was the one with the gold foil and thompson was the one who came up w/ the plum pudding model
"Inferred that atoms have a small, dense, positive nucleus" hint - this was a result of the gold foil experiment
C
good, so __B___ Believed that electrons move in specific orbits __A___ Discovered electrons __D___ Believed that atoms could not be broken into smaller parts ___A__ Experimented with cathode rays __C__ Inferred that atoms have a small, dense, positive nucleus ___A__ Developed plum-pudding model __C___ Shot particles at gold foil
Explain the progression of ideas from a variety of scientists that led to the current understanding of atomic theory.
this is a very bare-bones and I would recommend fleshing this out but: Dalton, used ideas about the atoms from the Greeks (particularly Aristotle), thought atoms were indivisible, every atom in a particular element is identical in composition JJ Thompson - plum pudding, cathode ray experiments, discovered charge of electron Rutherford - gold foil, model where atom had dense center w/ mostly empty space outside the center Bohr - came up w/ orbit model and the idea of quantized electron energy, positions of electrons could only be on integer energy levels Schrodinger: quantum atom model where electrons are de-localized in electron clouds instead of orbits
I would try to put this into coherent sentences, make sure to fill in approximate dates, do any additional clarification/research to help explain certain things in more detail, etc.
Each person should probably have its own short 2-5 sentence paragraph
did that, i added more details and dates.
hoose one example from the historical development of the atomic theory, and use it to illustrate how models are used in the process of forming conclusions from scientific experiments.
well, I think the Rutherford gold experiments are good example - since he wanted to test a hypothesis about the structure of the atom, had very unexpected results (the atom had a dense center that reflected particles), and had to come up with a better model to describe his results, which is an example of how he used a model to form a conclusion about the structure of the atom
something like that
I got the idea let me reword it :)
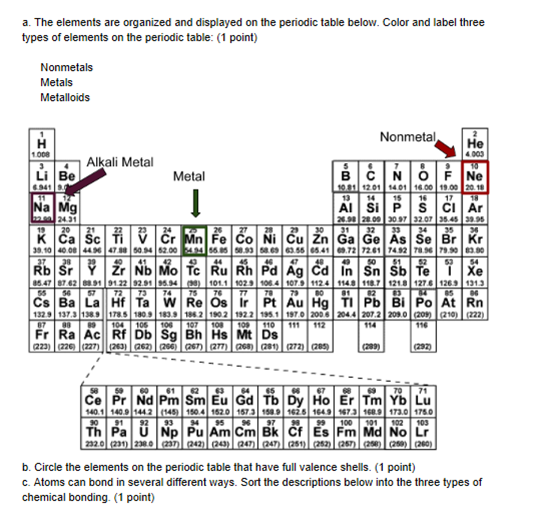
I am not sure about the phrasing for a) but it seems like they want you to color three regions not just three elements, the metal, nonmetal, and metalloid regions (just have to look up a periodic table and color the areas)
for b) full valence shells => last group, where helium and the noble gases are
okay and c
I can't see below where it says "sort the descriptions below"
Atoms can bond in several different ways. Sort the descriptions below into the three types of chemical bonding.
may I see the descriptions please
do you have any guesses/ any ideas where to start?
like, for "two atoms share electrons" do you remember which one this is?
covalent
good, what about 'two oppositely charged ions"?
ionic
good, what about metal + nonmetal?
you kinda just have to memorize this, it's ionic
any ideas for "delocalized electrons + positive ions"?
metallic
good two nonmetals -->?
ionic
nonmetals form covalent bonds w/ each other (just have to memorize this)
electron transfer between atoms -->?
metallic
close but not quite when ions are formed, they gain/lose electrons, right? and when ions bond w/ each other, electrons are transferred from the anion to the cation---> making ionic the better answer
that leads us into d) ---> metals lose electrons, and if a neutral metal loses electrons is the new charge positive or negative?
neg
electrons are negative charge so if the negative charges are going away, what's the charge of the remaining atom?
positove
good, that's it for d
When you hold a rectangular object, how does the area of the side that is resting on your hand affect the pressure and the force that the object exerts
P = F/A, force of gravity is constant for an object, so the area determines pressure
this is the answer right, there wouldn't be any numbers
yeah, we aren't given any numbers so we can't do any calculations
What are four types of energy that might make up the internal energy of a substance?
internal
heat chemical
I'm not 100% sure what they want as the answer here but I'd say translational kinetic energy, vibrational KE, rotational KE, potential energy from intermolecular forces
Yeah i wasn't sure either the question is very vague
(I don't know whether they want you to consider the macroscopic vs microscopic perspective)
anyway I should probably sleep I have an 8am tomorrow goodnight
See ya
Join our real-time social learning platform and learn together with your friends!