Given that the specific heat of water is 4.18 kJ/(kg·°C), how much heat does it take to raise the temperature of 3.5 kg of water from 25°C to 55°C?
q = mc * deltaT where m = mass c = specific heat deltaT = change in Temperature
3.5*4.18*20
292.6
check the change in temperature again, 55 - 25 is 30 not 20
3.5*4.18*30 = ?
438.9
good does your teacher care about significant figures (sig figs) or nah? b/c it would be 440 kJ if we round to significant digits
I can put both :p
sure
What is the mathematical equation for calculating the heat needed to change the temperature of a substance? Identify each variable and its units.
q = mc * deltaT, explanations are at the top
q = mc * deltaT where m = mass c = specific heat deltaT = change in Temperature
yup, any thoughts on units?
mass would be SI
[ok, so they gave you kg in the problem, but usually we would use grams for mass] so T is celcius m is grams specific heat = J/(g·°C)
oh, yeah, q is heat which is joules
Sooo, q = mc * deltaT where m = mass c = specific heat deltaT = change in Temperature T is celcius m is grams specific heat = J/(g·°C)
yes
Define specific heat capacity and latent heat of fusion. Specific heat capacity is the heat that is required to raise the temperature of the unit mass of a given substance. Latent heat of fusion is energy released or absorbed, by a body or a thermodynamic system, during a constant-temperature process that is specified in some way.
not bad but for latent heat of fusion it should be mentioned that this is the heat required to convert a substance from its solid to its liquid form (fusion)
Specific heat capacity is the heat that is required to raise the temperature of the unit mass of a given substance. Latent heat of fusion is energy that is released or absorbed during a constant-temperature process that is pinpointed in some way. Also, the heat in a latent heat of fusion is the heat required to convert a substance from its solid to its liquid form
good
How do particle motion and temperature change as a material absorbs heat?
Particle motion and temperature both increase as a material absorbs heat.
good
A physics student is sitting at the beach. She feels the warm Sun shining on her back, the warm sand under her feet, and a cool breeze blowing in from the water. Identify each type of heat transfer she is experiencing.
warm Sun shining on her back- radiation warm sand under her feet- cool breeze blowing in from the water-
well there are only two other types of heat transfer, conduction and convection, which one would be conduction and which one would be convection
warm Sun shining on her back- radiation warm sand under her feet-convection cool breeze blowing in from the water-conduction
other way around, conduction requires contact so that would be feet touching the sand
a. Match the term to its correct description. (1 point) A. Convection B. Radiation C. Thermal conductor D. Thermal insulator E. Conduction _____ A substance that transfers heat poorly _____ Heat transfer through direct contact of particles _____ Heat transfer through motion of a fluid _____ Heat transfer through electromagnetic waves _____ A substance that transfers heat well
Heat transfer through motion of a fluid is convection
good
Heat transfer through electromagnetic waves is radiation
yup
A substance that transfers heat well is conduction
that's not quite right, you're thinking of conductor
__D___ A substance that transfers heat poorly __E___ Heat transfer through direct contact of particles __A___ Heat transfer through motion of a fluid __B___ Heat transfer through electromagnetic waves __C___ A substance that transfers heat well
awesome
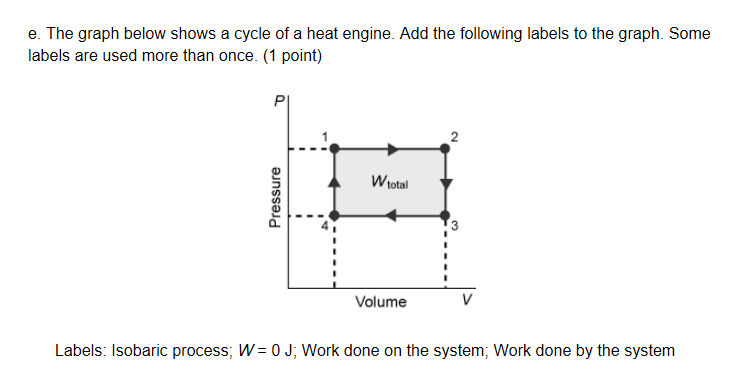
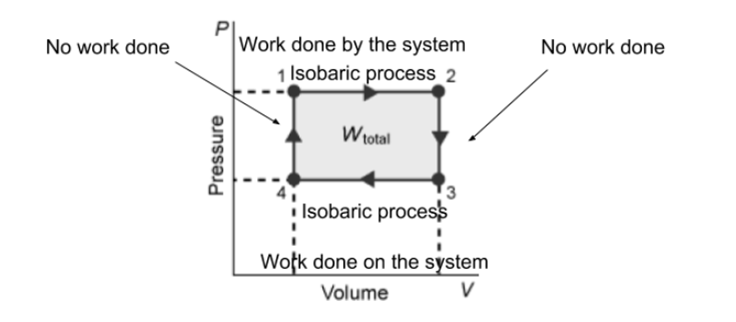
work = P*deltaV, where would work be 0?
the bottom
not quite, the volume is decreasing there so work would be nonzero where is the volume constant?
Would it be this
good, so the volume is constant on the left and right sides on the shape what about "isobaric process"? where is the pressure constant?
Um on the left
hm, not quite, if you compare points 1 and 4 see how the pressure is different? that means the left side is a process with non-constant pressure where on the graph is the pressure the same across both points?
top and bottom
awesome, so that would be your isobaric processes so, for work done on system, that means the volume must be going down, where would that be?
left side
notice how the volume is the same on the left side between points 1 and 4
volume is on the x-axis, which side on the graph shows a decreasing x-value?
the other side because points 1 and 4 show it going up
that's pressure not volume
|dw:1521646953811:dw|
|dw:1521646958611:dw|
what about work done by the system? that would be increasing volume, so which side?
opposite of the decreasing
good, so the top side and that's it
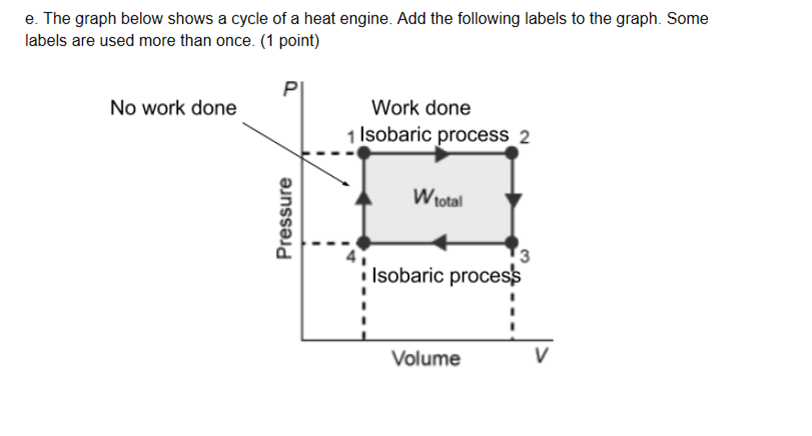
Something like this right
good, it's a bit incomplete atm, ex. no work done needs to be on the right side to also, for "work done" you need to specify whether the system is doing work or having work done on it
*on the right side too
the top side is expansion, thats work done (by) system not on system work done on system = bottom side
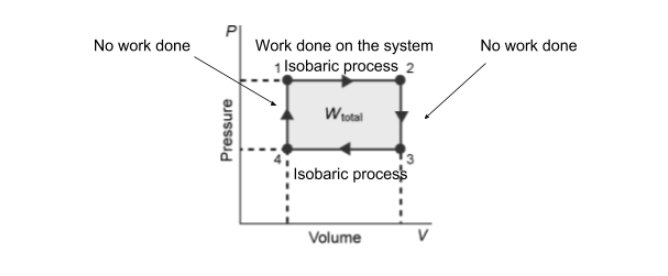
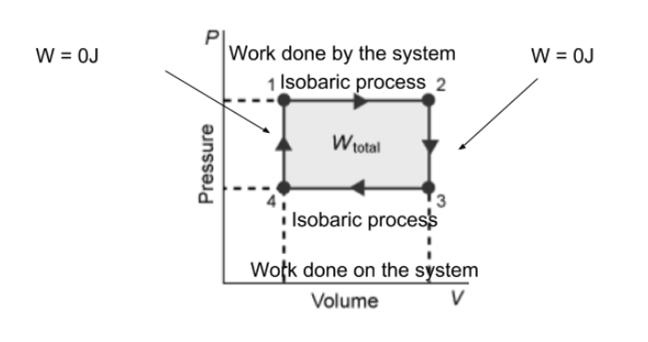
good as a minor issue the instructions say to label it as "W = 0J" not "no work done" so just change those labels and you'll be set
good, that's it
you're given e = W/Qin, work and heat-in are given in the problem, find e
e = W/Qin e=300/600 e=0.5
good, efficiency is often a percentage so we could report this as 50%
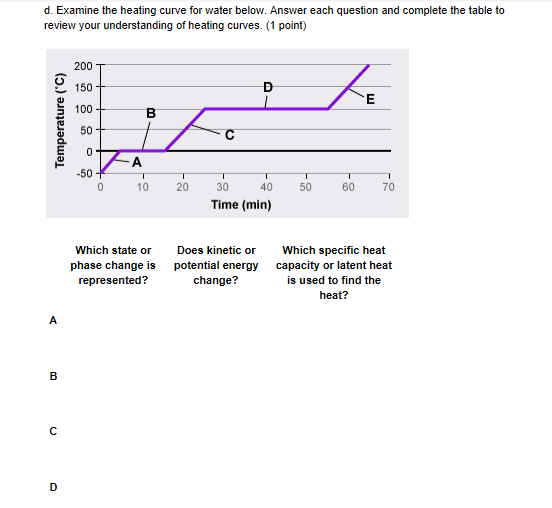
A is ice
good what about KE? does it change?
b is freezing/melting ice or water
no
for A) yes the KE does change (since heat is being put in + temp is changing) B) no since temp isn't changing
A is ice Yes kinetic or potential energy does change B is freezing/melting ice or water No kinetic or potential energy doesn't change because the temperature is not changing C is water Yes kinetic or potential energy does change D is boiling/condensing water or gas Yes kinetic or potential energy does change
E is gas No kinetic or potential energy does not change
check D and E again for KE changes
D is boiling/condensing water or gas No kinetic or potential energy does not change E is gas Yes kinetic or potential energy does change
good, what about the last column, for each letter which heat capacity would you use?
hint - for the phase changes it would be "latent heat of ______" whatever phase change it is and for the parts where the temperature changes it would be "specific heat capacity of ____" whatever phase its in right now
A. Specific heat capacity of solid B. Latent heat of C.Specific heat capacity of liquid D. Latent heat of E.Specific heat capacity of gas
for water, what would the names for its solid/liquid/ and gas forms be?
ice water and um
water vapor
for the phase change names, what's the name for a substance turning from solid to liquid, and then from liquid to vapor?
melting and evaporation
good but in chem/physics we usually call these fusion/vaporization so latent heat of melting/ latent heat of vaporization for B + D
A. Specific heat capacity of solid B. Latent heat of melting C.Specific heat capacity of liquid D. Latent heat of vaporization E.Specific heat capacity of gas
replace solid/liquid/gas with the names of their forms for water replace melting with its scientific name, fusion
A. Specific heat capacity of solid B. Latent heat of fusion C.Specific heat capacity of liquid D. Latent heat of vaporization E.Specific heat capacity of gas
\(\color{#0cbb34}{\text{Originally Posted by}}\) @Vocaloid *** replace solid/liquid/gas with the names of their forms for water *** replace melting with its scientific name, fusion \(\color{#0cbb34}{\text{End of Quote}}\)
A. Specific heat capacity of ice B. Latent heat of fusion C.Specific heat capacity of water D. Latent heat of water vapor E.Specific heat capacity of gas
D is latent heat of vaporization E is specific heat capacity of water vapor
A. Specific heat capacity of ice B. Latent heat of fusion C.Specific heat capacity of water D. Latent heat of vaporization E.Specific heat capacity of water vapor
good
Join our real-time social learning platform and learn together with your friends!