MCAT Mini-Tutorial: Understanding the Periodic Table

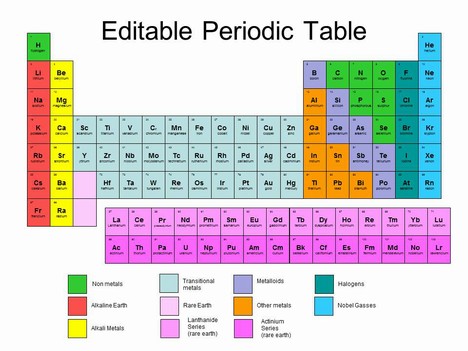
\({\bf{Basic~Layout:}}\) - period: rows - group: columns, elements in a group tend to share chemical properties - group 1: alkali earth (red on this diagram, excludes hydrogen), very reactive - group 2: alkaline earth metals (yellow), reactive but less so than group 1 - transition metals: d-block, light blue, partially filled d orbitals allow them to react w/, combine w/ other elements - rare earth elements: f-block, pink, first row is actinides and second row is lanthanides - group 7: halogens, very reactive - group 8: noble gases, non-reactive - transition metals: the blue-ish "staircase" between the transition metals and the nonmetals \({\bf{Classifying~Elements}}\) - metals: conductive, ductile, malleable, solid at room temp. - nonmetals: nonconductive, brittle, gaseous at room temp - metalloids: intermediary properties between metals and nonmetals \({\bf{Periodic~Trends:}}\) - atomic radius: 1/2 the distance between adjacent nuclei > decrease across period (more protons, same energy level, stronger effective nuclear charge) > increase down group (increase of shielding from lower energy levels) - first ionization energy: energy required to remove electron in gaseous state > increases across period (more protons, same energy level, stronger effective nuclear charge) > decrease down group: (increase of shielding from lower energy levels, easier to remove electrons) > subsequent ionization energy: increases as more electrons are removed (removing electrons increases the effective nuclear charge per remaining electron) - electron affinity: energy change of gaining an electron in gaseous state > same trends as ionization energy - electronegativity: affinity for electrons > same trends as ionization energy > metals have low EN, nonmetals have high EN, noble gases are not given EN since they are not reactive misc: - francium has lowest EN and largest radius - cesium is the most reactive
Anyway, that's the end of my tutorial, I hope it was a helpful resource. Source material is the 2nd Edition Barron's Prep book for the new MCAT
Join our real-time social learning platform and learn together with your friends!
