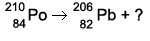
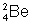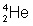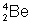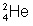Complete the following radioactive decay problem. (see attachment)
And the attachment is?
Alright so when doing this problem, both sides of mass and electrons must match. What do you think I mean by this
I get that part I just don't get which element to use
Which one do you think it is?
it's either option 3 or option 4
First, let's add both sides of the top number (mass number) What do you get on both sides?
The top number is going to be 4
What about the bottom? Always remember that the top number and bottom number (mass number and number of electrons) always match on both sides.
the bottom will be 2
So which one of the answer choices would contain 4 mass and 2 electrons
options 2 and 3
Correct. Do you have a periodic table with you?
I can pull one up
Oh well. Check the periodic table and see what element has 2 electrons. That will be your answer.
thank you for the help
Join our real-time social learning platform and learn together with your friends!