Orgo II Question about reactivity of aromatics
@Michael @BRAINIAC sorry to be a pain but could you take a look at this whenever you get a chance? :S
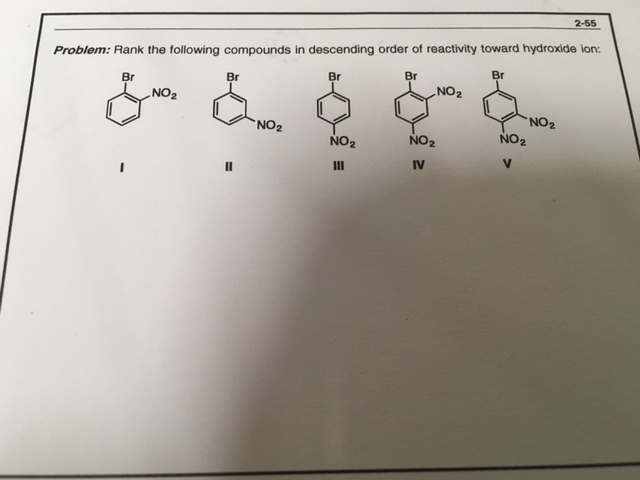
The intermediate formed in these reactions would be meisenheimer complex which would have a negative charge at the site where the hydroxide ion attacks.
So if you have some electron withdrawing groups at the ortho/meta positions they'd stabilise the intermediate and increase the rate of rxn
The mechanism followed is \(S_NAr\)
not para? does that mean structures I and III take precedence over the others?
Oops that should be *para over there instead of meta
oh then structure IV is the most reactive I guess?
Yep
between II and V does the meta group hinder the reaction in any way?
wait
oh, duh, since V has a para group then V takes priority over II
Yeah + I don't think that the meta group would interfere much.. because no2 isn't that big
my proposed solution: II, then I and III should be about the same, b/c they only have 1 o/p group each, then V, then IV?
wait
the problem says descending order so just flip that
I just talked to my professor and he says that V takes priority over II because of inductive effects of the nitro?
1 > 3 Because the size isn't a problem and the no2 on the ortho would also use the -I effect
\(\color{#0cbb34}{\text{Originally Posted by}}\) @Vocaloid I just talked to my professor and he says that V takes priority over II because of inductive effects of the nitro? \(\color{#0cbb34}{\text{End of Quote}}\) Thats correct But the main reason why 5>2 would be the -m of the para group
-m > -i ->Priority order
alright, thank you
np :-)
Join our real-time social learning platform and learn together with your friends!