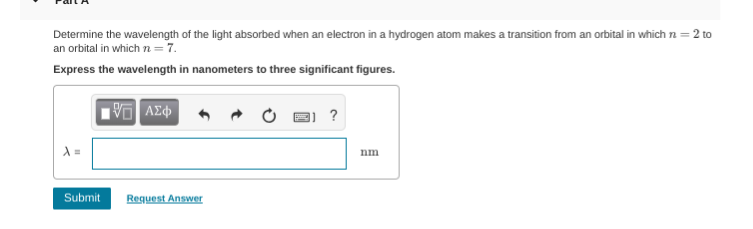
3.97 x 10^-7 ?
it wants the answer in nanometers yours is currently in meters, so simply multiply by 10^9
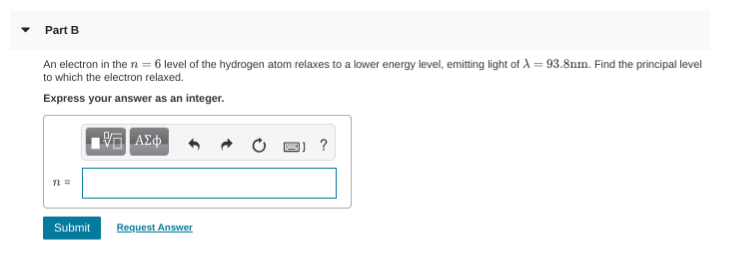
How do you find the principal level
ah so you are given ni = 6 and the wavelength you'd use the rydberg equation 1/lambda = R(1/nf^2 - 1/ni^2) to find nf
make sure to convert 98.3nm to meters first since the rydberg constant is in meters
1/(9.83x10^10) = (1.097x10^7)(1/nf^2 - 1/36)
wait
okay
should be -10 on that exponent 1/(9.83x10^-10) = (1.097x10^7)(1/nf^2 - 1/36)
I dont know why Im not getting a -10 ... oh well
ah should be -8
93.8nm --> divide by 10^9 --> 93.8e-8 whoops
but anyway 1/(9.83x10^-8) = (1.097x10^7)(1/nf^2 - 1/36) should give you your nf value
I thought it was
I thought it would be divived by 10^-9
ah, not quite, putting a negative exponent in the denominator --> end up with a positive exponent
if it helps to think of it this way, 93.8 nm is only a very small portion of a meter so we would expect the result to be very small thus we would want to divide by 10^9 not 10^(-9)
|dw:1538005835592:dw|
but what about the nano part,,, isnt nano 10^-9
10^(-9) would be converting m to nm we want the other way around this time
oh okay I see. Since it is like the inverse the signs would change
like, there's two ways to write the conversion factor 1 m = 10^9nm or 1 nm = 10^(-9) m so both of these conversion factors would work you just gotta be careful about how to use them
1 m = 10^9nm gives us 98.3nm/10^9 or 1 nm = 10^(-9) m gives us 98.3nm*10^(-9) both give the same result using different conversion factors
1/(9.83x10^-8) = (1.097x10^7)(1/nf^2 - 1/36) should give you your nf value anyway chucking this into a calculator gives 1 as your nf value, so n = 1 is the principal energy value and thus your sol'n
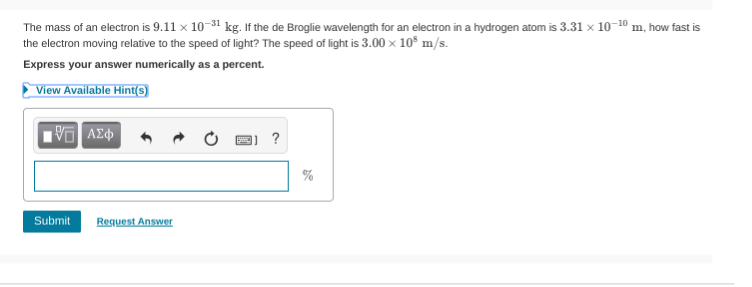
Okay, so I got 3.31 x 10^-10
for this one
I’m on my phone so I can’t really check this easily But it wants it as a percentage of the speed of light So you’d solve for v in the de brogue equation then do v/c * 100
so v is 2.20x10^6 its a given... so (2.20x10^6)/(2.998x10^8) * 100
i got 7.34 x 10^15
Hm that doesn’t sound right Anyway i need to workout but I asked someone to take over for me >>
OKay =)
Hi zarkam, I'm trusting you're using the correct numbers here. If that's true, then I think you should get around 0.734 % as your answer.
To see why this is true, consider that (10^6)/(10^8)*100 simplifies to 1. 10^6 * 100 = 10^8; 10^8/10^8 = 1 So, the only relevant calculation you have to do is 2.20/2.998. As you can see for yourself, that equals about 0.734
where is the 2.20 coming from
Haha, that's the number you gave my dude. But if we want to calculate the velocity of the particle from the information given, we can use the equation: \[\lambda=\frac{ h }{ p }\] where lambda stands for the de broglie wavelength of the particle, h stands for planck's constant, and p stands for the momentum of the particle (recall that momentum is equal to mass multiplied by velocity)
Could you explain what I did wrong here?
Hm, did you remember to convert the mass to kilograms when calculating momentum?
Nvm got it lol sorry
Ok, awesome
@SmokeyBrown I have a question> : What is the de Broglie equation used for?
like to find what?
So, the de Broglie equation describes the relationship between a particle's wavelength and its momentum (using Planck's constant) Basically, if you have the wavelength, you can find the momentum, and if you have the momentum, you can find the wavelength. Since momentum can be broken down into mass and velocity, you can also calculate these values, provided you have the other values required in the equation.
SO h wouldbe plancks.. v would be (velocity /speed) I'm confused with what m would be
I know it would be mass but in a problem like : Calculate the wavelength of an electron traveling with a speed of 3.21 × 105 m/s..... I don't know what the mass would be
Oh, for that question you'd just have to know that all electrons have approximately the same mass. According to Google, this value is 9.10938356 × 10^-31 kilograms
OKay so 2.27 x 10^-9?
?
uhhhhhhh yes
Sorry, it took a while for me to plug that into my calculator
Join our real-time social learning platform and learn together with your friends!

.png)