Mg(NO3)2?
Magnesium Nitrate?
Mg=Magnesium NO3=nitrate You're correct (:
BUt its NO NItrogen and Oxygen?
The NO3 is a polyatomic ion. Why do we know that? Because every compound has to be equal charges, and Nitrogen charge is -3 while Oxygen is -6, and that doesn't work because the charges aren't equal.
OH okay so NO3 is nitrate then
Yes, so you were correct!
NH4ClO3
Ugh I really wanna figure this out on my own
BUt this would be a polyatomic too?
Try it out! You were already right the first time for the first problem
Do you have to memorize the polyatomic ions, or no?
no
I mean I don't think kso
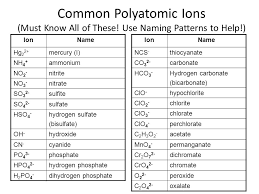
Alright, then use this picture for your help.
What chemistry are you in? Most of the times, regular high school chemistry doesn't force you to remember them.
Im in college chem
Using that polyatomics chart and what you know, try to form a formula.
general chemistry
Prepare to memorize it, just a side note. I'm in college level chemistry in high school, and they made me remember them.
Okay thanks for the chart i really appreciate it
and I got umm ammonium chlorate?
Of course, that chart is your drugs to passing chemistry
And yes, you are right
PbO would just be Lead oxide?
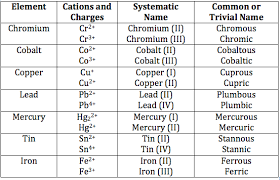
Partially right :P You see in the chart that Lead has multiple charges, right?
NO :/
Oh, it doesn't say! I'm sorry.
if you have like a chemistry go-to folder, add that polyatomic list and this one.
Now, looking at that chart you can see that Lead has multiple charges, am I correct>
Yup I see it
It has charge 2 and 4. Since it is common sence (as oxygen charge is -2 and lead must be 2 because the ratio is 1:1) that the lead is charge 2, we still have to show that Lead has the charge of two in this compound because it could be four.
Kind of confusing, I know. But to show that Lead has the charge of plus 2, you can add a parenthesis after Lead and add the roman numeral of how many charges they have.
So the actual answer would be Lead (ll) Oxide You were almost right ;P
its easier if you just draw it
Give the systematic name for the compound Mg(NO3)2.
Magnesium Nitrate?
for this one
Yep!
Fe2(SO4)3.
I know the first one is Iron (ii) but not sure if it is sulfate or sulfide
No, the iron has either a charge 2 or 3, and in this case, it is a 3 because |dw:1539491364144:dw|
Does that make sence?
A little
So it would be Iron (III) Sulfate?
Since the charge of one sulfate ion is -2, and there are three of them, the overall charge is -6. The other element always has to have the same opposite charge, so Fe2 must be 6. There are two, and so we divide it by two, and so the charge of one iron is 3. So the formula is Iron (lll) sulfate
OH i get it
barium oxide.
would be BaO
?
yes, thats correct
lead(II) phosphate is Pb^+2 P
Not quite. Remember the polyatomic ions, phosphate is PO4
oh right so Pb^+2 PO4
Lead is Pb, and charge is either 2 or 3. Phosphate is PO4, and charge is -3. To balance, we have to find a common multiplier for 2 and 3, which is 6.
Lead must be charge three because phosphate is 2.
Oh it always has to be in its balanced form
Yes
Pb^+3 PO2
:)
The formula for phosphate is PO4
SO is it 3Pb 4PO4
it would be Pb3(PO4)2 You were very close.
The charges have to be equal, Pb charge is 2, multiply by 3=6 Phosphate charge is -3, multiply by 2=-6 They are both equal.
is the 3 an expoonent?
No, there would be no exponents
\[Pb _{3}(PO _{4})_{2}\]
Join our real-time social learning platform and learn together with your friends!