Help
@lowkey
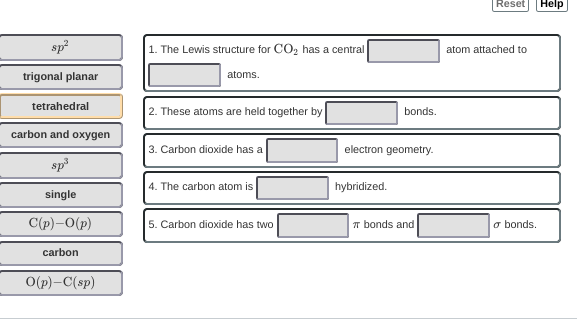
Alright, so do you think you can make a CO2 Lewis Structure for me in order to answer 1 and 2?
|dw:1540936352263:dw|
I know for the first blank, the central atom would be carbon
It's actually double bonds on the carbon and oxygen so O=C=O Carbon only has four electrons but wants four more so they can achieve that 8 electron atom that all atoms want. So they kind of force oxygen to share it with them.
well the first blank would be carbon regardless
You're right haha
for the second blank woudl it be carbon and oxygen?
So we established that the first blank would be carbon. Side question, do you know why it's a carbon in the middle not just because we drew it out?
because of the electronegativity
When making lewis dot structures, the atom with the least amount of electronegativity goes in the middle.
Dang it you smarter than me now :/
Yea, so your second answer is right. An alternative would be double bonds but there is no double in the key.
Then for the third question, it is asking about the geometry about the electrons.
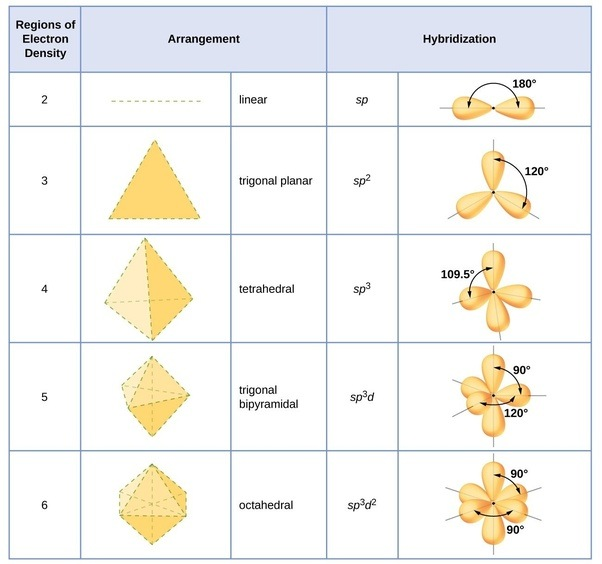
One thing that helped me out during chemistry was this sheet
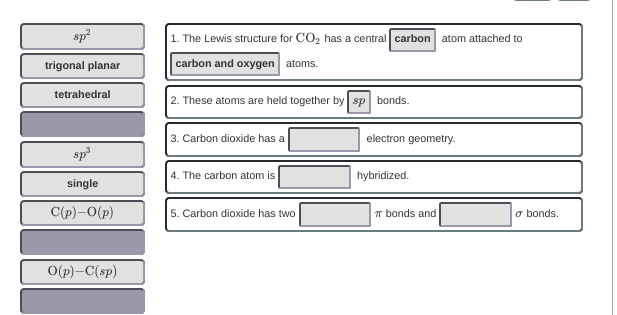
I can get rid of some answer choices, but number three answer is supposed to be sp or linear because there are no lone pairs and they contain double bonds, therefore making it a linear geometry, but there is no word from the bank :/
I don't know what it would be then :/
Hold on, lemme see the sheet.
okay
which sheet?
What choices can we rule out of the answers?
would it be sp bonds?
See, you're correct, I just don't see it in the choices :/
Actually forget it, can we work on this one instead
OH SP WAS CUT OFF
And I also do not know how to do the second picture actually );
Join our real-time social learning platform and learn together with your friends!