stumped
@Vocaloid
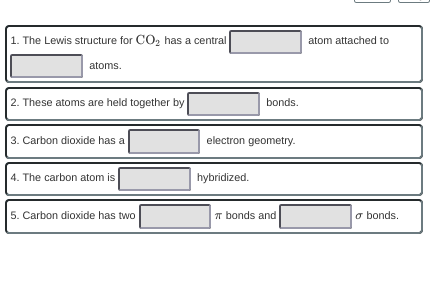
1. there's only two choices (C and O) the less electronegative atom goes in the center and the more EN atoms go on the outer edges so would C be the first blank or O?
c would be the first
for the second blank:
good, so carbon first blank and oxygen second blank
2. ideas about the bond type? it would either have to be covalent or ionic
covalent
*looks at answer choices* oh wait that's not one of the choices whoops I guess they're looking for either single or double. any attempts to draw the lewis structure?
it would be double bonded
good, so double
c in the middle with 2 o's with double bonds
linear for the next geometry
3. ideas for the geometry? 2 bonds, 0 lone pairs
good
4. hybridization, any ideas? if you are stumped try calculating the steric #
sp3
hm, not quite, steric number is 2 (C is bonded to 2 other atoms, plus 0 lone pairs) so sp
how do you determin the steric NUm again?
|dw:1540949586801:dw|
i'd have to think a bit about 5 but the pi orbitals should result from the overlap of p orbitals
yeah well pi bonds are always p
yeah that should be it
What is the hybridization of the central iodine atom in I3−?
sp3d
good that's what i got too
it says : Express your answer in the orbital notations s, p, d, and f.
so that would still be right
yes
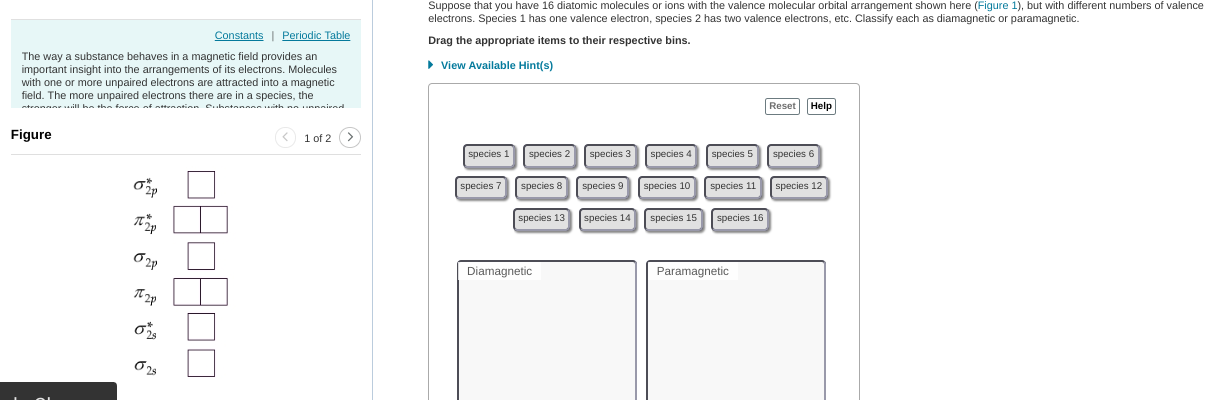
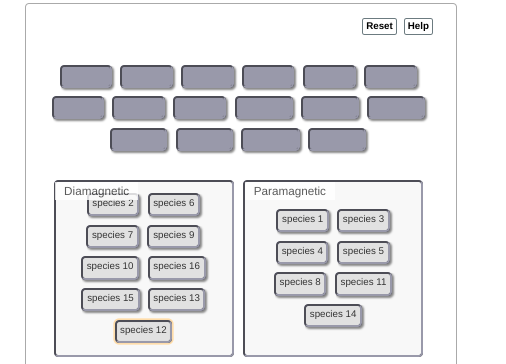
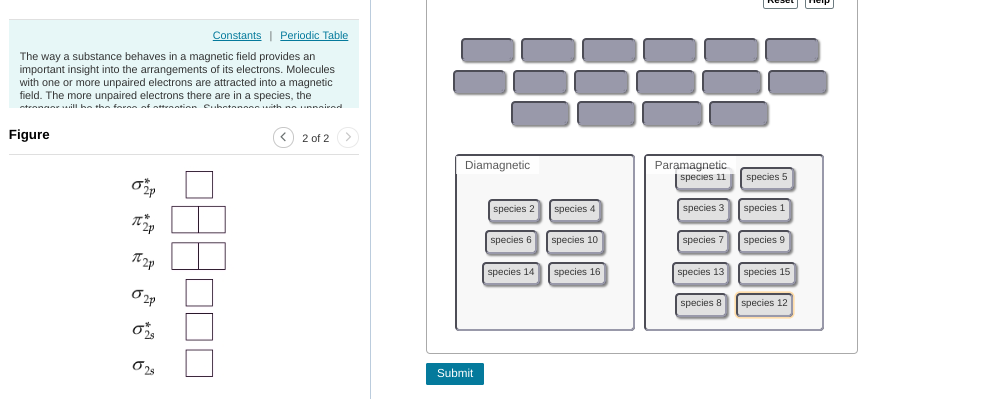
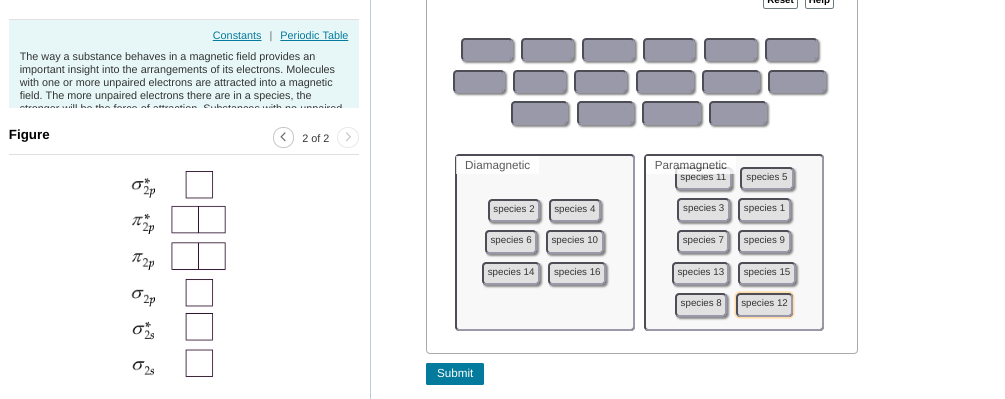
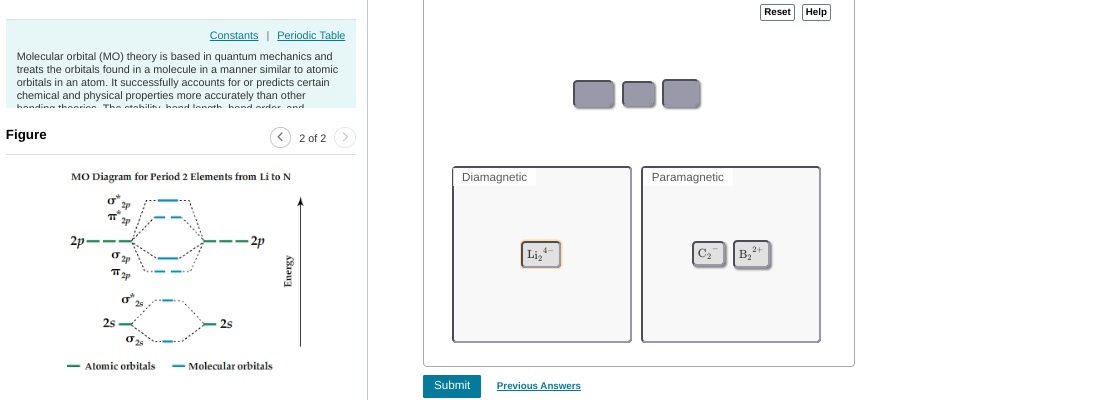
this can be tedious but you'd start with compound 1 (1 valence electron), put 1 electron in to the MO diagram. --> that one ends up unpaired so this is paramagnetic then you'd continue this process w/ the other compounds
need to re-do some of them for example, 6 is paramagnetic. you'd fill up the first 4 electrons but then the last 2 are unpaired b/c they occupy separate orbitals
as a hint: any odd number will be paramagnetic (if there are an odd number it's impossible for them all to be paired) for even numbers you have to consider on a case-by-case basis
8 should be diamagnetic (they all get paired up w/in the first four orbitals) but everything else is good
6 should be paramagnetic per our earlier reasoning (unpaired electrons in 2pi bonding) diamagnetic should contain: 2, 4, 8, 10, 14, 16; everything else is para
hm. :/
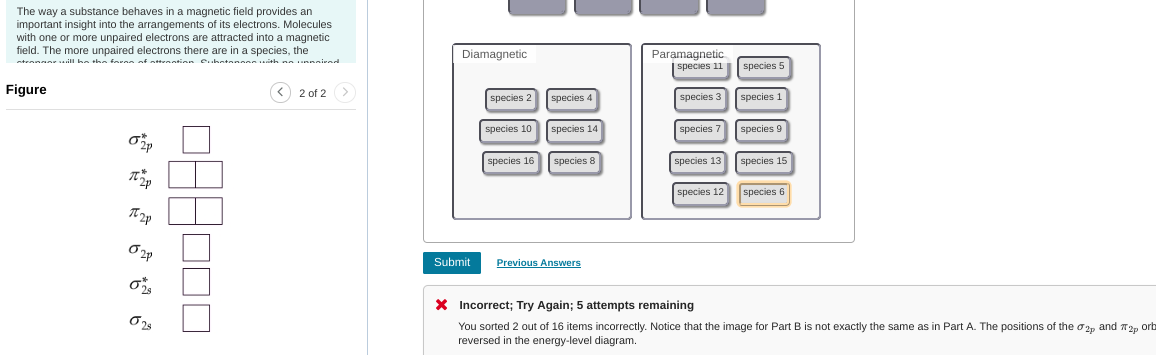
oh I see they wanted a different orbital diagram than the one they used before
yeah and thats what I based my previous answer off of
This
then yeah, your orig. should have been right
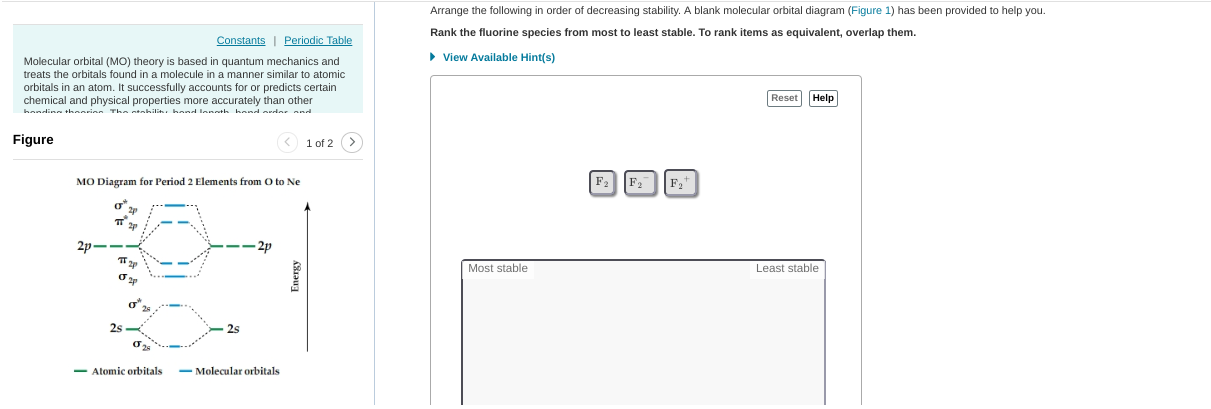
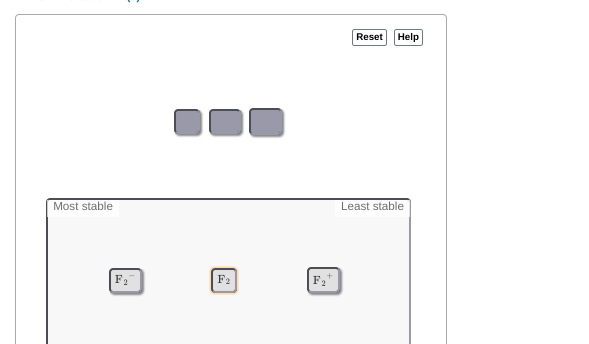
that's a good attempt but F2- fills up all of the anti-bonding orbitals and anti-bonding orbitals are not desired for stability. I'd put F2- at the bottom F2+ keeps the antibonding orbitals empty so it's the most stable F2 goes in between so basically just reverse the order you have already
anything with a * symbol (sigma*, pi*) etc. are antibonding orbitals
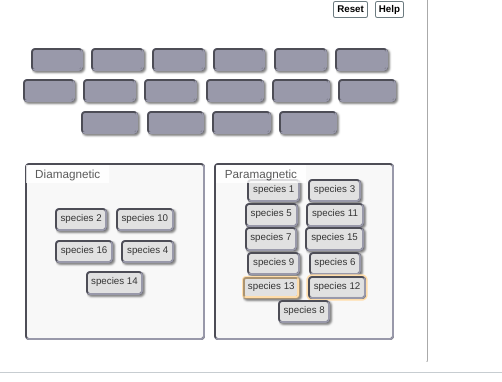
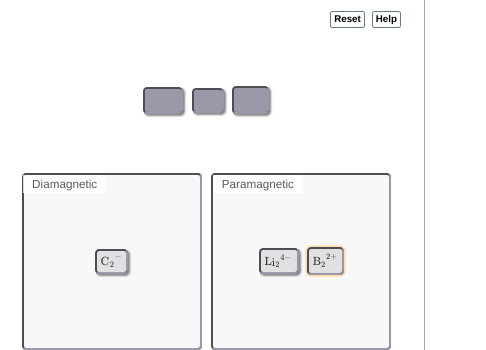
|dw:1540952522320:dw| not 100% sure on this but thinking the lithium might be diamagnetic
|dw:1540952536946:dw|
okay so lithium and carbon would be dia
I don't really feel super confident on these tbh
if I"m not mistaken B2(2+) has 8 total electrons, occupying the first four orbitals, therefore being dia
oh since each C has 6 electrons -- 12 electrons --> +2 because it's an anion --> 14 total electrons --> fills up the first 7 orbitals to be diamagnetic so if moving C2(2-) doesn't work idk where to go from there
so all three dia
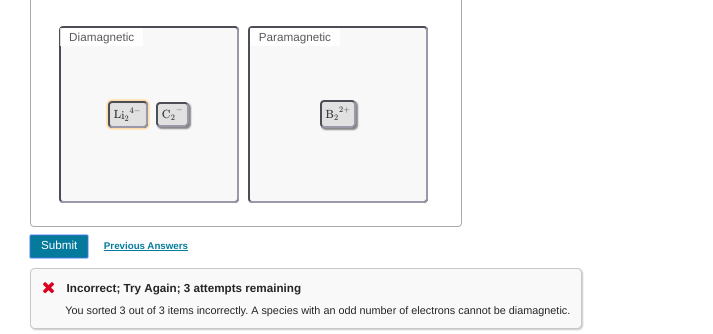
no, B2(2+ is still para
ugh now its 3 out of 3 incorrect
oh in that case that makes it super easy, just switch all the para's to dia's and all the dia's to paras
ohhh I see where I went wrong, there's a -1 charge on the carbon not a -2 charge >_>
got it ughh thankssss
Join our real-time social learning platform and learn together with your friends!