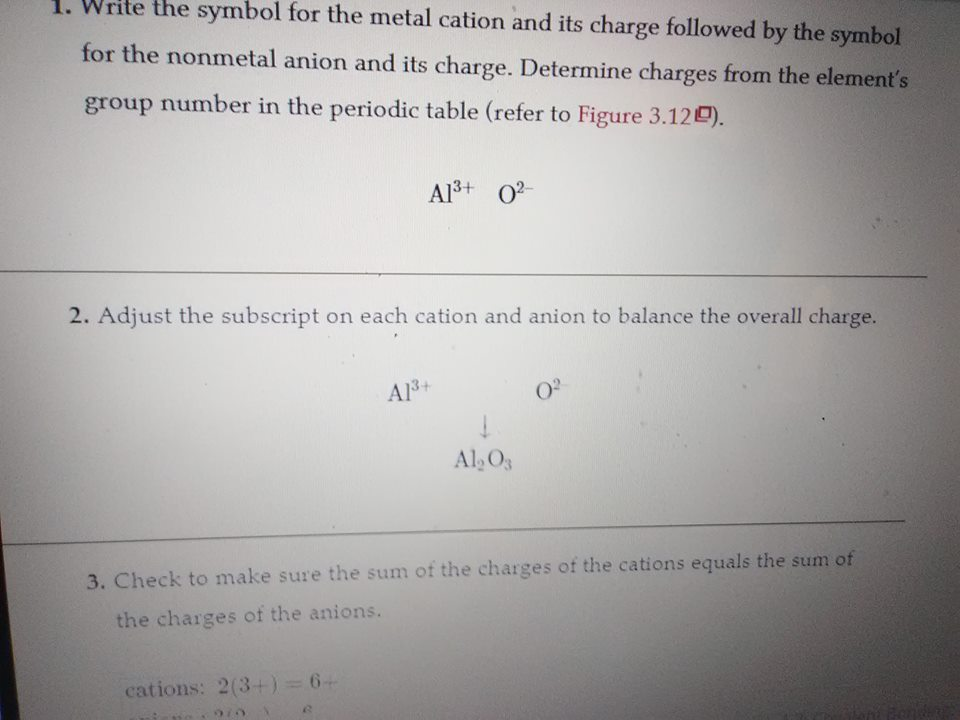Explanation?
Sodium must lose its one valence electron in order to have an octet (in the previous principal shell), while sulfur must gain two electrons to get an octet. Consequently, the compound that forms between sodium and sulfur requires two sodium atoms to every one sulfur atom—the formula is Na2S. The two sodium atoms each lose their one valence electron, while the sulfur atom gains two electrons and gets an octet. The Lewis model predicts that the correct chemical formula is Na2S
@Vocaloid
ON this as well: Calcium needs to lose its two valence electrons (to be left with an octet in its previous principal shell), while chlorine only needs to gain one electron to get an octet. Therefore, you must have two chlorine atoms for each calcium atom. The calcium atom loses its two electrons to form Ca2+, and each chlorine atom gains an electron to form Cl–. In this way, both calcium and chlorine attain octets.
I don't get what they mean by you need "..." for each "..."
"Sodium must lose its one valence electron in order to have an octet (in the previous principal shell), while sulfur must gain two electrons to get an octet. Consequently, the compound that forms between sodium and sulfur requires two sodium atoms to every one sulfur atom—the formula is Na2S." each sodium atom has to lose 1 VE to get an octet sulfur needs to gain two electrons to get the octet so if each sodium is donating 1 electron, sulfur needs 2 sodium atoms in order to get the full octet
same logic w/ calcium and chlorine
OKay but why does sodium need to lose one versus gain 7?
sodium has an extra 1 electron in its valence shell it could technically gain 7 but that would require a tremendous amount of energy, so it's more convenient to lose 1 instead
so where is the cutoff of whether an element should either like gain 5 or lose just the 3
like how do know its better to lose or gain
do you just look at it and see which would require the least amounto f energy and go with that
metals (left side of ptable) like to lose; nonmetals (right side of ptable) like to gain
Calcium needs to lose its two valence electrons (to be left with an octet in its previous principal shell), while chlorine only needs to gain one electron to get an octet. Therefore, you must have two chlorine atoms for each calcium atom.
Don't get this part: "Therefore, you must have two chlorine atoms for each calcium atom." Why would you need two chlorine atoms for each calcium atom?
"ON this as well: Calcium needs to lose its two valence electrons (to be left with an octet in its previous principal shell), while chlorine only needs to gain one electron to get an octet. " each chlorine atom needs 1 electron each calcium atom gives away 2 electrons so for each atom to have a full octet, two chlorine atoms need to donate 1 electron each, so calcium gets 2
if it's confusing to think about the actual atoms, try thinking about the charges instead each chlorine ion has a -1 charge; each calcium ion has a +2 charge, so to get to 0 you need 2 chlorines and 1 calcium (-1 * 2 +2 = 0)
Yeah it is easier this way
How do you know the charges
ptable
|dw:1541034222277:dw|
Write the symbol for the metal cation and its charge followed by the symbol for the nonmetal anion and its charge. Determine charges from the element’s group number in the periodic table (refer to Figure 3.12). Al3+O2–
alright any ideas? if aluminium is +3 and O is -2 how many of each do you need to get 0 charge?
so al is the metal and o is the nonmetal
yes
2 o's for every one al?
IDk its not balancing out :S
no, if you had 2O and 1Al that would be -2(2) + 3 = -1
easy question what's the least common multiple of 2 and 3?
oh we take the charges I forgot
9
6
**
good, 6 so the aluminiums have to sum to +6 and the oxygens have to sum to -6 see what you can get from that
0
good, but how many atoms of Al and O?
2 for al and 3 for o
good so you end up w/ Al2O3
oh okay so take the LCM and then determine how many of each we would need to get that LCM and then jut use that
yes
how would I Adjust the subscript on each cation and anion to balance the overall charge.
idk how your program works try shift underscore or something
I mean how would I do num 2
we did the calculations to determine that it would take 2 aluminium and 3 oxygen atoms to make a neutral molecule so you would write A2O3
oh okay so that woul dbe it for number 2
yes
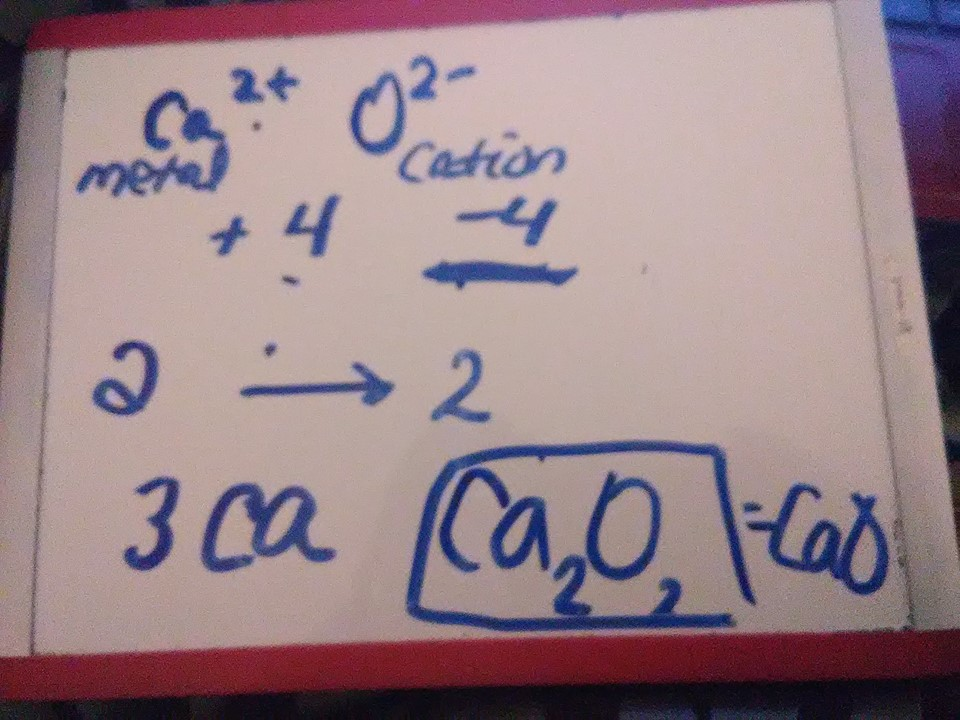
Here's another example: iF YOU could just check how I did
you have +2 and -2, these already cancel out to 0 so just CaO
So i basically just did it the long way
yeah it's fine, just make sure to reduce at the end if possible
Name the compound CaBr2. SOLUTION The cation is calcium. The anion is from bromine, which becomes bromide. The correct name is calcium bromide
WOuldn't the 2 subscript count as anything . I thought it would make it di- something
nah that's only for nonmetal + nonmetal compounds
ohhh
HOw do you know all this :S
Like you have it all memorized and its soooo much material
idk I took honors chem + AP Chem + freshman chem so it's been reinforced a lot i gues
oh right, and your a chem major right
yes
Name the compound Ag3N. This would be Silver nitride
yup
so this is the metal and nonmetal ones right?
yes
The full names for compounds containing metals that form more than one kind of cation have the form:
what does this mean: "That forms from more than one kind of cation"
well, you know how alkali metals/alkaline earth metals have set charges? like Ca will always make Ca2+? there are some elements (mostly transition metals) that can have different charges depending on what they're attached to Iron is a good example, it can be Fe2O3 (+3 charge) or FeO (+2 charge)
right
so these compound make the form metal(charge in roman numerals) nonmetal+"ide" suffix so using the other example, FeO (+2 charge) ---> iron(II) oxide
so its basically any compound that has cation elements right
then you would put the parentheses
like only for cations
yeah it's generally only cations that do this, I can't think of any anions that do this
oh i see, you mean any cations that have more than one charge participate in this
yeah. unfortunately you just have to know which cations do this but here are the major ones |dw:1541040955862:dw|
Join our real-time social learning platform and learn together with your friends!