need help finding percent yield here is the chart that I need to fill out: https://prnt.sc/mr1dcv So I know that in order to find the percent yield I first need to find the actual yield and also the theoretical yield. I believe. But im having some trouble finding these....
This terminology seems oddly familiar to something I am doing. Were you not given a formula to follow?
No I don't think so. I was only given a formula to find the percent yield. percent yield = (actual yield/theoretical yield) * 100)
I mean I was given this but im not sure how much it helps 2 water + sugar + lemon juice = 4 lemonade
The terminology I was used to was measured and true value, where measured was what something was found to be and true was what it actually was...
hmm
oh... im not sure if its the same thing or not.
Okay so the percent yield is how much the reaction produced vs. how it was supposed to produce if given all correct enviroments. For example, if we were given hydrogen and chloride and we found 27 grams of the gas, while the instructions say you were supposed to get 30, the percent yield is 27/30*100
can you upload the doc on here?
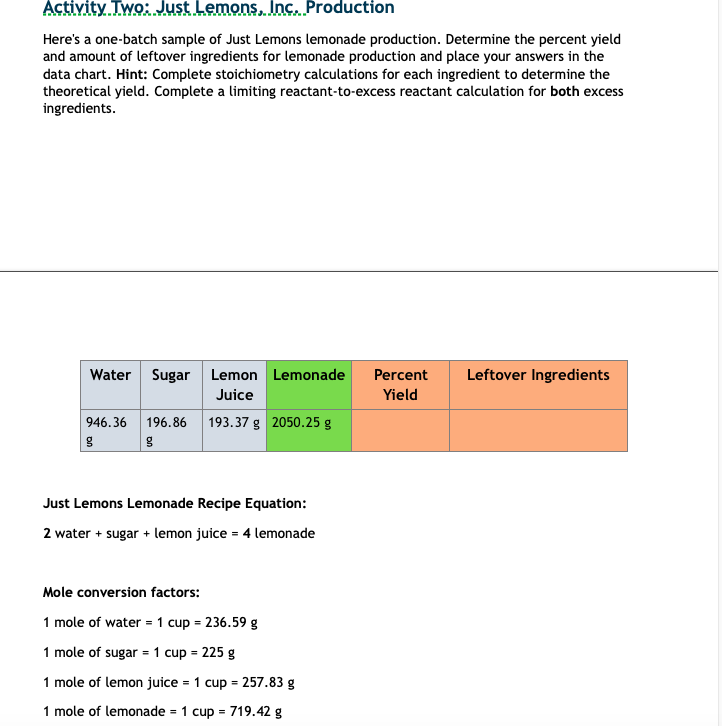
it won't let me attach it.... but it says this: here's a one-batch sample of Just Lemons lemonade production. Determine the percent yield and amount of leftover ingredients for lemonade production and place your answers in the data chart. Hint: Complete stoichiometry calculations for each ingredient to determine the theoretical yield. Complete a limiting reactant-to-excess reactant calculation for both excess ingredients.
then it shows the chart from above
@shadow convert link for me
and it also shows this: 1 mole of water = 1 cup = 236.59 g 1 mole of sugar = 1 cup = 225 g 1 mole of lemon juice = 1 cup = 257.83 g 1 mole of lemonade = 1 cup = 719.42 g so I have no idea how to go about this
doesnt make sence. is there a formula for the lemonade making steps
@studydeep The "Attach File" function isn't working?
not for me... it might be on my end but im not sure. and I think you mean this lowkey.... 2 water + sugar + lemon juice = 4 lemonade
wait what
have you learned any of this? idk seems confusing
yes i have when is this due
i kinda have to go but if i was given time i could finish this out for you
its kinda past due.... only by like a few days... but yeah whenever you have time. I would appreciate any help!
aww im sorry haha
i really need the whole problem picture though. can you screenshot it or take a picture then upload it using the 'attach file' tab below this text box
Yeah I can do that... its basically what I copied and pasted but if this helps at all.
I'll do this until tomorrow at this time, ok?
Thank you! I really do appreciate it!
ya ur welcome
now go get some rest or sum, ill figure this out
I meant to get to this earlier but you are given grams of each each ingredient and must convert to moles. you can accomplish this by dividing grams/molar mass for example, you have 946.36g water and you are given 1 mole of water = 1 cup = 236.59 g, so moles of water = (946.36) / (236.59) = 4 moles of water now, you can go to the equation 2 water + sugar + lemon juice = 4 lemonade this means 2 moles of water make 4 moles of lemonade, so basically, ignoring the amount of sugar and lemon juice you have, you could theoretically make 8 moles of lemonade from 4 moles of water
however, since you have multiple ingredients, you must also take into account how much sugar and lemon juice you have. simply convert grams of sugar into moles of lemonade using this same process, and repeat again with grams of lemon juice to moles of lemonade. at the end of this process you will have three quantities 1. how many moles of lemonade you could make with the amount of water you were given 2. how many moles of lemonade you could make with the amount of sugar you were given 3. how many moles of lemonade you could make with the amount of lemon juice you were given whichever is the smallest of these three quantities is the limiting factor. this is a bit hard to grasp but think of it this way: you could have 1000 tons of water and sugar, but if you only have 1 cup of lemon juice, you can only make 1 cup of lemonade.
now, the amount of lemonade you can make with your limiting factor is your *theoretical yield* percent yield, as you stated before, is 100(actual yield/theoretical yield)% you are given the actual yield of lemonade as 2050.25, so calculating percent yield should be easy once you have your theoretical
as for the last step, leftover ingredients, you would have to backtrack and calculate how much sugar, lemon juice, and water you actually ended up using to make the lemonade, then just subtract that from the original quantities of ingredients given
anyway I know this is a lengthy explanation so feel free to ask about anything you're still not 100% sure about
Okay I think I see what you mean here. Could you help a little more with the three quantities to find the theoretical yield "1. how many moles of lemonade you could make with the amount of water you were given 2. how many moles of lemonade you could make with the amount of sugar you were given 3. how many moles of lemonade you could make with the amount of lemon juice you were given" I keep trying to convert but I feel like each time im getting the wrong answer, the whole conversion part is the most confusing for me.
2 water + sugar + lemon juice = 4 lemonade tells you how to convert from moles of water, sugar, and lemon juice into lemonade. 2 moles of water = 4 moles lemonade 1 mole of sugar = 4 moles lemonade 1 mol lemon juice = 4 moles lemonade
Oh I see. Then you said "smallest of these three quantities is the limiting factor." and then " the amount of lemonade you can make with your limiting factor is your *theoretical yield* " is lemon juice the smallest? im not sure.
anyways just let me know once you have time. This assignment is a little bit past due so im trying to hurry while also trying to understand the material :) thank you for helping!!
Join our real-time social learning platform and learn together with your friends!