orgo mechanism question
https://imgur.com/a/bud6tQs I know the final product will be C b/c of anti-M addition and enol to keto tautomerzation but looking for some assistance on the mechanism
Please do not comment unless you are actually familiar with the subject material and are making a serious attempt at a solution.
I would try and help, but the link is blocked on my computer.
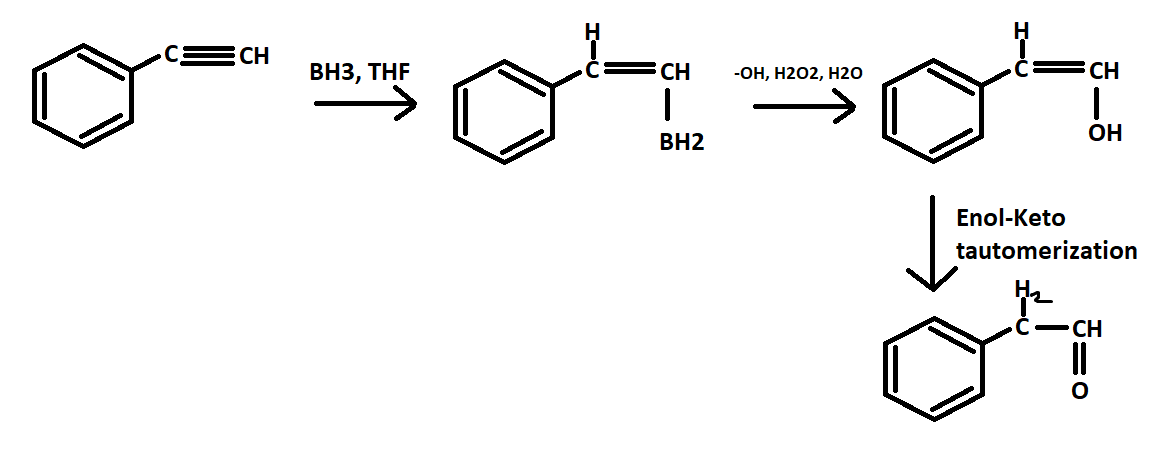
This is a hydroboration of an alkyne. Which will form your enol and then you have an enol to keto tautomerization because the keto form is more stable.
|dw:1564156400165:dw| I couldn't find anything in particular for alkynes, but it should be similar to alkenes. Alkyne: https://www.khanacademy.org/science/organic-chemistry/alkenes-alkynes/alkyne-reactions/v/hydroboration-oxidation-of-alkynes This was more over the mechanism of the tautomerization https://www.masterorganicchemistry.com/2011/07/08/reagent-friday-bh3-borane/ Here, you can see how BH3 forms that ring structure, and one H goes to the more substituted carbon (since it's more EN) leaving BH2 on the other carbon. In their example, that gets rid of the alkene, so presumably in an alkyne, we would be left with an alkene which would form our enol in the next step Underneath that, they provide the mechanism for how OH replaces the Boron
thank you C:
My pleasure :)
Join our real-time social learning platform and learn together with your friends!