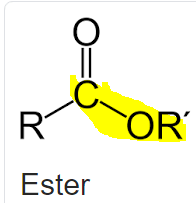
do esters contain ethers?
does the C-O-R not count as an ether?
You could argue that they both have an ether linkage but for practical purposes they’re considered distinct functional groups because the -OR behaves differently in both groups
So to put it more simply, the image you posted would not be considered an ether
yeah, thanks, got a really misleading question asking to tick which functional groups are present in a molecule picture. I mean, it's there, but you would not call what is there an ether, when naming it.
omg, just realised theres also a ketone option. great
|dw:1566564708530:dw| this is it btw
out of carbonyl, ketone, ester, ether and carboxylic acid, i have carbonyl and ester ticked, but now i'm doubting myself
oo
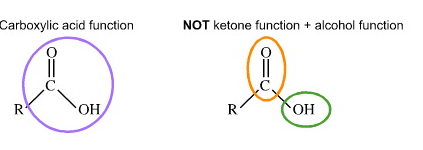
Yeah the one you drew would still be considered an Esther For it to be a ketone or aldehyde there can’t be an oxygen attached to the same carbon
ketone had got to be with connected to only Cs yea?
C's*
Yeah
lol i swear if i get this wrong
wait.. ethers are R-O-R, does a C with no H on it count as an alkyl group?
Probably not, alkys are typically defined as groups with only C and H
an alkyl is a functional group of an organic chemical that contains only carbon and hydrogen atoms. So no I dont think that would count
yeah, it's such a stupid question tho...
oops didnt realize vocaloid answered already
dw, i need all the help i can get
"if the letter A is in CAT, is it still A?" bruh....
i'm gonna go with carbonyl and ester
Yeah that’s probably what I’d go with too
Don’t worry this stuff confused the heck out of me when I was taking Orgo
it's part of a small test that you do and submit once, and get absolutely no feedback on, just if you get a question wrong it's boom!, 2 marks lost
wow thats crappy
everyone googles for it anyways
I suppose good luck.
|dw:1566568580894:dw| what groups does this have?
are there any choices with this one?
Join our real-time social learning platform and learn together with your friends!