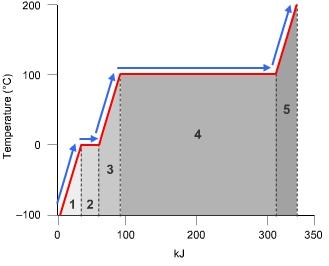
In which labeled portion of the curve would you use the heat of fusion to calculate the heat absorbed? 1 2 3 4
here is the graph
This, as you can see, is an enthalpy diagram, and it is asking for the heat of fusion. This is the heat required for a phase change, from liquid to solid, or basically the melting/freezing point. Which do you think shows a phase change for this substance (it looks like water) from liquid to solid/solid to liquid.
4 i would say
since it is large
4 is heat of vaporization, not heat of fusion.
Also it has nothing to do with being large. The length of those flat lines has to do with the energy being used to actually form/break the intermolecular forces. That's why it's flat.
ohhhh okay , hmm then 2 ?
A hint is that this diagram is basically the enthalpy diagram for water. Water freezes/melts at 0°C, so the heat of fusion is at 0°C, correct?
yes 2
ohh okay, yes that makes more sense. it would then be 2 ! awesome (: im glad i can make sense of this! thanks
NP (:
Join our real-time social learning platform and learn together with your friends!