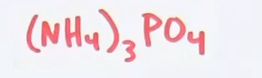Can someone help me on some chemistry please? *I'll post the images*
This is for the last question on my edpuzzel
hmm ok give me a min
Okie
It was the 2nd one, thank you though. I got all the rest correct
It's okay, at least you tried. Thank you again
mhm
Just so you know how it's done in the future: \[(NH_{4})_{3}PO_{4}\] in chemistry, the number near the bottom right (subscript) of an element only applies to that element. so the 4 on H ---> 4 hydrogens per 1 NH4 the 4 on the O ---> 4 oxygens per O, etc. now, if there's a subscript next to something in parentheses, it applies to the whole thing inside the parentheses so the 3 on (NH4)3 means 3 separate units of NH4
so, putting it all together \[(NH_{4})_{3}\] ---> 3 nitrogen atoms \3*4 = 12 hydrogen atoms PO4 ---> 1 phosphorus atom, 4 oxygen atoms in total: 3 nitrogen atoms, 12 hydrogen atoms, 1 phosphorus, 4 oxygens now, go to the periodic table, and add up the masses. start with the mass of 1 nitrogen atom (14.01 g/mol), multiply by 3 ---> 42.03 g/mol from nitrogen and repeat with the other atoms, then add up the result
Thank you voca, that helped explain it better
Join our real-time social learning platform and learn together with your friends!