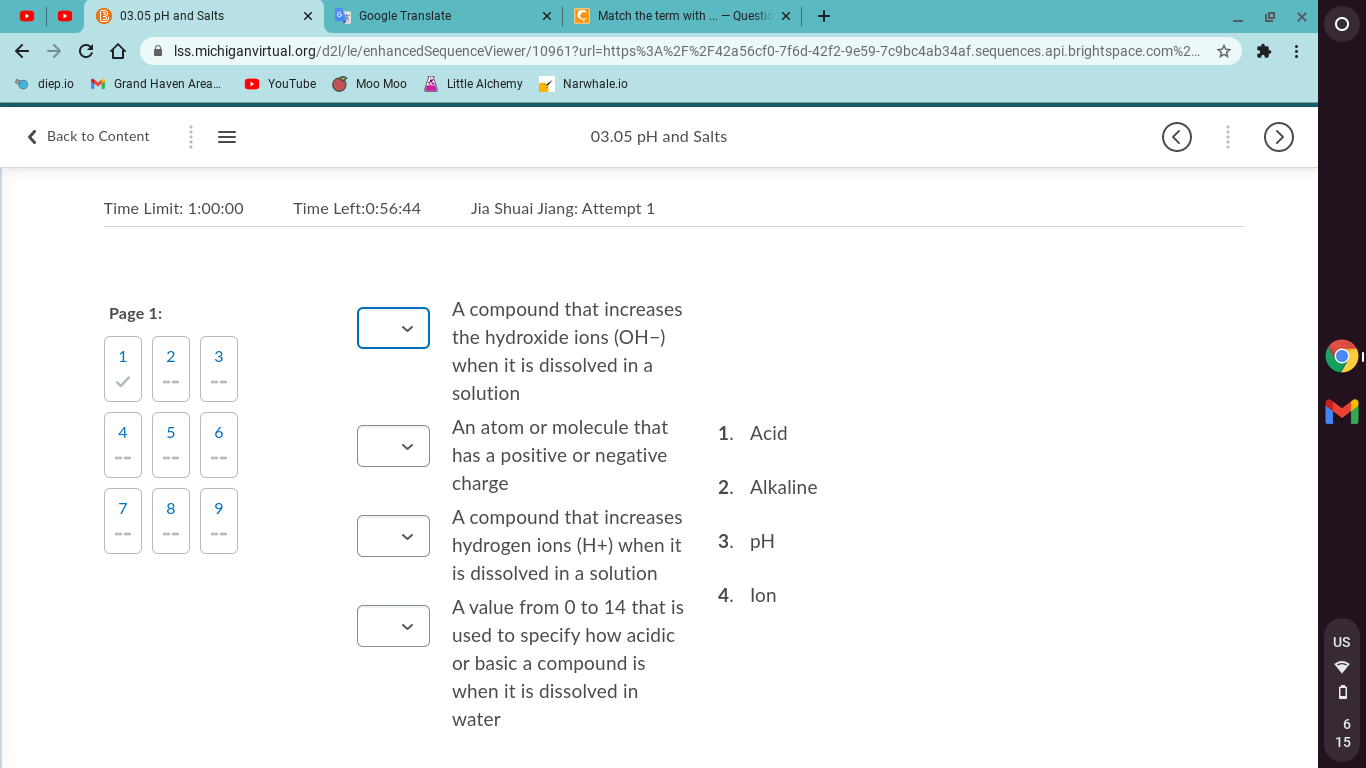
Match the term with its description.
help pls
Let's start with one of the easier definitions: pH. What all do you know about pH?
okay
It is a scale, helping us to determine how acidic or basic something is. Do you know what range numbers that scale uses?
no
Well, take a look at your choices and see which one best matches the description I gave you. After we figure out where pH goes we can move on to another term
the first one?
Nope, pH is a scale or a range of values, not a compound or atom of any sorts! Those would fall into the pH scale, though
ok so Acid has more H+ so Alkaline has more what?
Oh okay
do you need help with the rest?
yes pls
ok
ok if electrons make ions and you need electrons to determine a positive or negative charge...
I need it fast
ion..
I gotta go
i'll list it in order
okay thx
metal and fan Have a great day
thx
Join our real-time social learning platform and learn together with your friends!