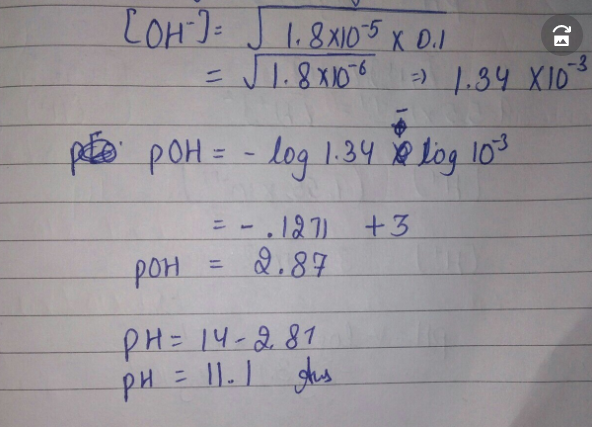Can someone plz help me with science...idk how to do this Determine the pH of 0.10 M NH3 solution. NH3 is a weak base with a Kb equal to 1.8 × 10-5. (Round answer to nearest whole number.)
is there any answer choices to go with it
no
it is 11 if rounded or 11.42 , 0.68%
we have to find ka as kb is given it can be found by using pKA plus p KB is equal to pKW as we know it is 14 from there you'll get pKa as 9.25 from that a is equal to 5.6 into 10 power minus 10 from there you can find the h + Ion concentration and substitute in pH you get the pH of H Plus
i hope that helps you to figure it out
tht just confused me even more lmao
Ok lemme break it down for you
it is 11 if rounded or 11.42 , 0.68% this is what i got
ok thank u....
oh okay thank u sm
your welcome
Join our real-time social learning platform and learn together with your friends!