Any particular element can always be identified by its _____. atomic mass number of neutrons number of electrons number of protons
what do you think the answer could be?
any clue of what the answer might be?
it would be atomic mass number of neutrons number of protons atomic number.
Just going to copy-paste my previous reply from your other post since it seems like you never got to the final correct answer
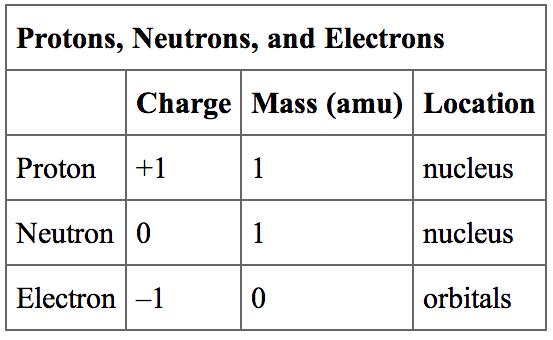
So an element is made up of a certain number of protons, neutrons, and electrons. Remember- protons are positively charged, neutrons are neutral, and electrons are negatively charged. Let's say an element has 8 protons. (This is oxygen by the way which you can tell from the periodic table). So a normal oxygen would have 8 protons, 8 neutrons, and 8 electrons. If we change the number of electrons, we're going to form ions. If we add an electron to an element, it'll become negatively charged. And similarly, if we take away an electron, it'll become positively charged. If we change the number of neutrons, we're going to form isotopes. An isotope is the same element (so it has the same number of protons) but it has a different number of neutrons.
To understand what atomic mass is, let's take a step back It is what it sounds like. We're just talking about the mass of the element. So the element is made up of protons, neutrons, and electrons. But what is the mass of a proton, neutron, or electron? Since electrons are tiny, their mass is really close to 0. But protons and neutrons are larger so their mass plays a major role. And we define their mass as 1 amu. So when we're trying to calculate the atomic mass, it's the average of all the different isotopes based on how common it is to find in nature
So with all that information, @sky18 what should the answer be?
Join our real-time social learning platform and learn together with your friends!