The nuclear equation is incomplete. \(\begin{matrix}1 \\ 1\end{matrix}\text{H} + \begin{matrix}15 \\ 7\end{matrix}\text{N} \to~ ? + \begin{matrix}4 \\ 2\end{matrix}\text{He}\) What particle completes the equation? Superscript 12 Subscript 6 Baseline M g Superscript 11 Subscript 5 Baseline B Superscript 11 Subscript 5 Baseline N a Superscript 12 Subscript 6 Baseline C
In order to balance a nuclear equation. We need to make sure that the mass is the same on both sides. And also the total nuclear charge has to be the same. https://www.youtube.com/watch?v=8hnhhBQBQy4
looks hard
so to find the missing particle the top number is the mass number the bottom number is the atomic number the sum of it should be equal on both sides so let's say our missing particle is \( \begin{matrix}1 \\ 1\end{matrix}\text{H} + \begin{matrix}15 \\ 7\end{matrix}\text{N} \to~ \begin{matrix}x \\ y\end{matrix}\text{?}+ \begin{matrix}4 \\ 2\end{matrix}\text{He}\) and then we figure out what the element is based on what `y` or the atomic number is based on periodic table so 1 + 15 = x + 4 what is the mass number of the particle? what is x? and 1 + 7 = y + 2 what is the atomic number of the particle? what is y? then use the periodic table using the atomic number to find out what the element is for example if the atomic number is 5, look at the periodic table and you'll see it's B and if it's 8 is O
let me know what you get @TheerealShawnteee
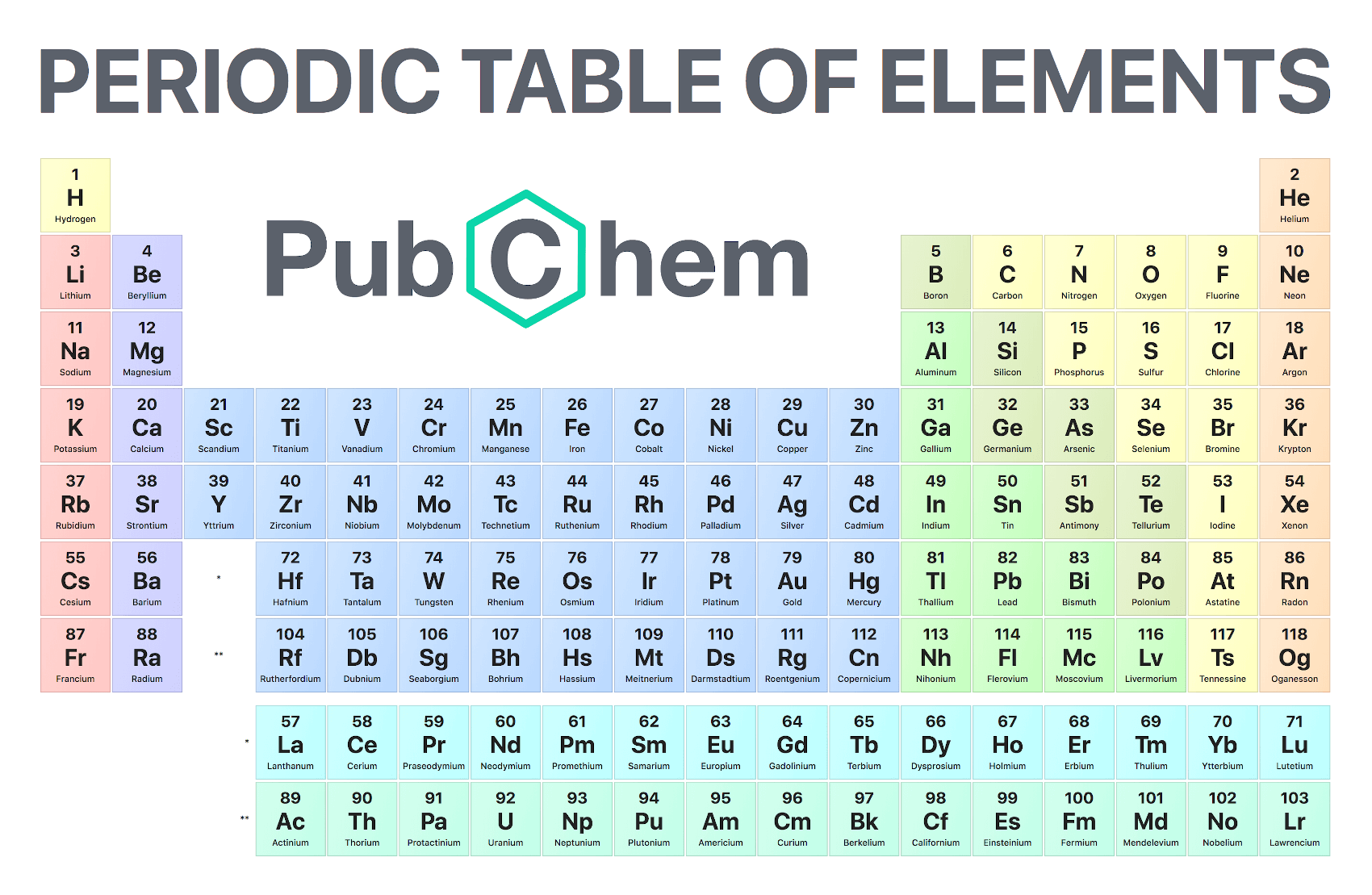
Here's a periodic table to help you
lord jesus
i just cheated on brainly
that's sad
Join our real-time social learning platform and learn together with your friends!